What are the common ICD 10 codes?
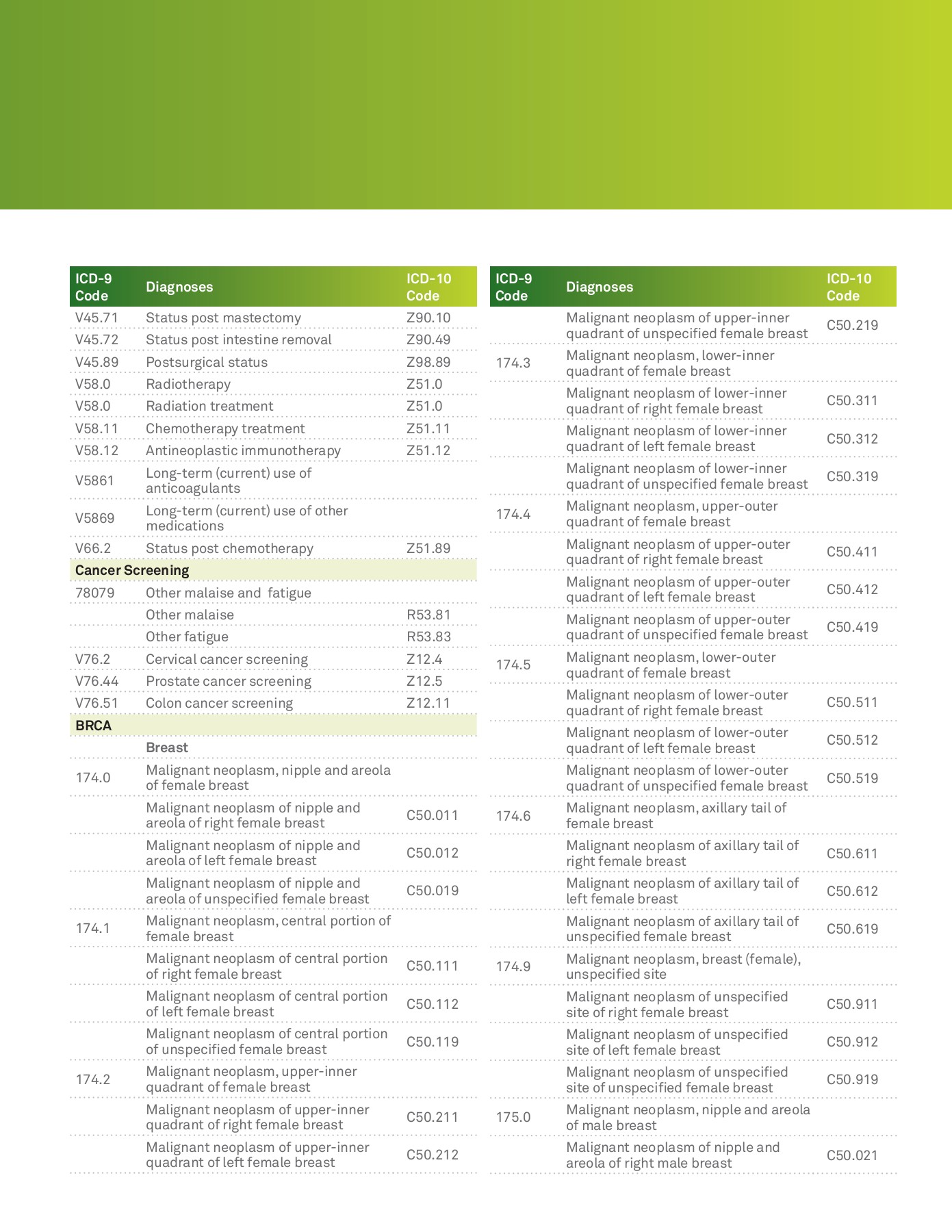
ICD-9-CM and ICD-10-CM Common Codes for BRCA1 and BRCA2 ICD-9 Code ICD-10 Code Breast 174.9 Malignant neoplasm, breast (female), unspecified site C50.911 Malignant neoplasm of unspeci˚ed site of right female breast C50.912 Malignant neoplasm of unspecified site of left female breast C50.919
What are the new ICD 10 codes?
Family history of carrier of genetic disease. 2016 2017 2018 2019 2020 2021 2022 Billable/Specific Code POA Exempt. ICD-10-CM Diagnosis Code Z15.01 [convert to ICD-9-CM] Genetic susceptibility to malignant neoplasm of breast. Brca1 gene mutation positive; Brca2 gene mutation positive; Genetic marker brca1; Genetic susceptibility to breast cancer; Genetic …
What is the difference between ICD 9 and ICD 10?
Oct 01, 2021 · Z15.01 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes. The 2022 edition of ICD-10-CM Z15.01 became effective on October 1, 2021. This is the American ICD-10-CM version of Z15.01 - other international versions of ICD-10 Z15.01 may differ.
Where can one find ICD 10 diagnosis codes?
The ICD-10-CM code Z15.01 might also be used to specify conditions or terms like brca1 gene mutation positive, brca2 gene mutation positive, breast cancer genetic marker of susceptibility positive, breast cancer genetic marker of susceptibility positive, breast cancer genetic marker of susceptibility positive , li-fraumeni syndrome, etc.
Can Z15 01 be a primary diagnosis?
Genetic susceptibility indicates that a person has a gene that increases the risk of that person developing the disease. Codes from category Z15 should not be used as principal or first-listed codes.
What does BRCA1 positive mean?
A positive test result means that you have a mutation in one of the breast cancer genes, BRCA1 or BRCA2, and therefore a much higher risk of developing breast cancer or ovarian cancer compared with someone who doesn't have the mutation. But a positive result doesn't mean you're certain to develop cancer.Aug 12, 2021
What type of gene is BRCA1?
BRCA1 and BRCA2 are two genes that are important to fighting cancer. They are tumor suppressor genes. When they work normally, these genes help keep breast, ovarian, and other types of cells from growing and dividing too rapidly or in an uncontrolled way.
What does Z12 31 mean?
For example, Z12. 31 (Encounter for screening mammogram for malignant neoplasm of breast) is the correct code to use when you are ordering a routine mammogram for a patient. However, coders are coming across many routine mammogram orders that use Z12.Mar 15, 2020
What is the difference between BRCA1 and 2?
Differences Between BRCA1 and BRCA2 Both mutations increase the risk of ovarian cancer, as well as pancreatic cancer. A BRCA1 mutation can also increase the risk of cervical, uterine, and colon cancer, while BRCA2 can increase the likelihood of stomach, gallbladder, and bile duct cancer, plus melanoma.Jun 10, 2013
Is BRCA1 or 2 worse?
Which Gene Mutation is Worse, BRCA1 or BRCA2? By age 70, women BRCA1 carriers have a slightly higher risk of developing breast cancer than BRCA2 carriers. Also, BRCA1 mutations are more often linked to triple negative breast cancer, which is more aggressive and harder to treat than other types of breast cancer.Jun 18, 2020
What chromosome is BRCA1 on?
BRCA1 is located on chromosome 17. Mutations in the gene are transmitted in an autosomal dominant pattern in a family. Since it was clear that not all breast cancer families were linked to BRCA1, studies continued and in 1994, scientists discovered another gene (similar to BRCA1), and named it BRCA2.
What is the function of BRCA1 and 2?
BRCA1 is a pleiotropic DDR protein that functions in both checkpoint activation and DNA repair, whereas BRCA2 is a mediator of the core mechanism of homologous recombination.Dec 23, 2011
What chromosome is BRCA1 and BRCA2 on?
Abstract. Two cancer susceptibility genes, BRCA1 on chromosome 17q12-21 and BRCA2 on chromosome 13q12-13, are thought to be responsible for approximately 80% of families containing multiple cases of early-onset female breast cancer.
What is the ICD-10 code for screening for osteoporosis?
Z13.820Encounter for screening for osteoporosis Z13. 820 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes.
What is ICD-10 code for osteoporosis?
0 – Age-Related Osteoporosis without Current Pathological Fracture. ICD-Code M81. 0 is a billable ICD-10 code used for healthcare diagnosis reimbursement of Age-Related Osteoporosis without Current Pathological Fracture.
What is Z12 11 ICD-10?
Z12. 11: Encounter for screening for malignant neoplasm of the colon.May 1, 2016
What is the ICd 10 code for breast cancer?
Z15.01 is a billable diagnosis code used to specify a medical diagnosis of genetic susceptibility to malignant neoplasm of breast. The code Z15.01 is valid during the fiscal year 2021 from October 01, 2020 through September 30, 2021 for the submission of HIPAA-covered transactions.#N#The ICD-10-CM code Z15.01 might also be used to specify conditions or terms like brca1 gene mutation positive, brca2 gene mutation positive, breast cancer genetic marker of susceptibility positive, breast cancer genetic marker of susceptibility positive, breast cancer genetic marker of susceptibility positive , li-fraumeni syndrome, etc. The code is exempt from present on admission (POA) reporting for inpatient admissions to general acute care hospitals.#N#The code Z15.01 describes a circumstance which influences the patient's health status but not a current illness or injury. The code is unacceptable as a principal diagnosis.
What is the name of the disease where certain cells in the breast become abnormal and multiply uncontrollably to form
Breast cancer Breast cancer is a disease in which certain cells in the breast become abnormal and multiply uncontrollably to form a tumor. Although breast cancer is much more common in women, this form of cancer can also develop in men.
Is Z15.01 a POA?
Z15.01 is exempt from POA reporting - The Present on Admission (POA) indicator is used for diagnosis codes included in claims involving inpatient admissions to general acute care hospitals. POA indicators must be reported to CMS on each claim to facilitate the grouping of diagnoses codes into the proper Diagnostic Related Groups (DRG). CMS publishes a listing of specific diagnosis codes that are exempt from the POA reporting requirement. Review other POA exempt codes here.
General Information
CPT codes, descriptions and other data only are copyright 2020 American Medical Association. All Rights Reserved. Applicable FARS/HHSARS apply.
CMS National Coverage Policy
Title XVIII of the Social Security Act, Section 1833 (e) states that no payment shall be made to any provider of services or other person under this part unless there has been furnished such information as may be necessary in order to determine the amounts due such provider or other person under this part for the period with respect to which the amounts are being paid or for any prior period..
Article Guidance
This Billing and Coding Article provides billing and coding guidance for Local Coverage Determination (LCD) L36499, BRCA1 and BRCA2 Genetic Testing. Please refer to the LCD for reasonable and necessary requirements.
ICD-10-CM Codes that Support Medical Necessity
It is the provider’s responsibility to select codes carried out to the highest level of specificity and selected from the ICD-10-CM code book appropriate to the year in which the service is rendered for the claim (s) submitted.
ICD-10-CM Codes that DO NOT Support Medical Necessity
All those not listed under the “ICD-10-CM Codes that Support Medical Necessity” section of this article.
Bill Type Codes
Contractors may specify Bill Types to help providers identify those Bill Types typically used to report this service. Absence of a Bill Type does not guarantee that the article does not apply to that Bill Type.
Revenue Codes
Contractors may specify Revenue Codes to help providers identify those Revenue Codes typically used to report this service. In most instances Revenue Codes are purely advisory. Unless specified in the article, services reported under other Revenue Codes are equally subject to this coverage determination.
Document Information
CPT codes, descriptions and other data only are copyright 2020 American Medical Association. All Rights Reserved. Applicable FARS/HHSARS apply.
CMS National Coverage Policy
This LCD supplements but does not replace, modify or supersede existing Medicare applicable National Coverage Determinations (NCDs) or payment policy rules and regulations for BRCA1 and BRCA2 genetic testing services. Federal statute and subsequent Medicare regulations regarding provision and payment for medical services are lengthy.
Coverage Guidance
Notice: It is not appropriate to bill Medicare for services that are not covered (as described by this entire LCD) as if they are covered.
What is the BRCA test?
Unless you deal with the tests for breast cancer (BRCA), estrogen receptor (ER)/progesterone receptor (PR), and human epidermal growth factor receptor 2 (Her2) — or have gone through a diagnosis of breast cancer or know someone who has — you may not be familiar with them and their use in diagnosing and treating breast cancer.
How is breast cancer coded?
Breast cancer can be coded by accounting the stage of the cancer. Breast cancer staging is based on the TNM system developed by the American Joint Committee on Cancer from seven key pieces of information:
How to code breast cancer?
Breast cancer can be coded by accounting the stage of the cancer. Breast cancer staging is based on the TNM system developed by the American Joint Committee on Cancer from seven key pieces of information: 1 Size of the tumor (T) 2 How many lymph nodes has the cancer spread to (N) 3 Has the cancer metastasized to other sites (M) 4 Is ER positive (ER) 5 Is PR positive (PR) 6 Is Her2 positive (Her2) 7 Grade of cancer (G)
What is triple negative breast cancer?
Triple Negative. Triple negative breast cancers (ER-/PR-/Her2-) occur in 10-20 percent of all breast cancers (and are more common in BRCA1 mutations). Women with triple negative breast cancer have tumor cells that do not contain receptors for ER, PR, or Her2. These tumors are treated with a combination of therapies, including surgery, chemotherapy, ...
What are the genes that are involved in the production of tumor suppressor proteins?
BRCA1 and 2 are genes that have been identified in the production of tumor suppressor proteins. These genes are integral to repairing damaged deoxyribonucleic acid (DNA). Mutations of these genes increase the risk of breast and ovarian cancers. One study found that approximately 72 percent of women who inherit a BRCA1 mutation ...
What percentage of women inherit BRCA1?
One study found that approximately 72 percent of women who inherit a BRCA1 mutation and approximately 69 percent of women who inherit a BRCA2 mutation will develop breast cancer by the age of 80. The following CPT® codes can be used for BRCA1 and 2 mutation testing:
How are cancer cells graded?
Cancers cells are given grades based on how much the cancer looks like normal cells: Grade 1 cells are slower growing, well differentiated, and look more like normal breast tissue. Grade 2 cells are growing at a speed between grades 1 and 3, moderately differentiated, and look between grades 1 and 3 cells.
Why is a BRCA1 negative?
A negative test could be due to lack of inheritance of a BRCA1 or BRCA2 abnormality (true negative), due to testing an inappropriate gene (false negative). In some cases, false-positive results can arise due to the presence of a clinically insignificant polymorphism in one of the BRCA genes.
What is LCIS in breast biopsy?
Women with atypical hyperplasia of lobular or ductal origin and/or lobular carcinoma in situ ( LCIS) confirmed on biopsy with dense, fibronodular breasts that are mammographically or clinically difficult ...
What is a pseudoangiomatous stromal hyperplasia?
An UpToDate review on “Overview of benign breast disease” (Sable, 2016) states that “Pseudoangiomatous stromal hyperplasia -- Pseudoangiomatous stromal hyperplasia (PASH) is a benign stromal proliferation that simulates a vascular lesion. PASH may present as a mass or thickening on physical examination. The most common appearance on mammography and ultrasound is a solid, well-defined, non-calcified mass. The characteristic histologic appearance is a pattern of slit-like spaces in the stroma between glandular units. PASH can be confused with mammary angiosarcoma. If there are any suspicious features on imaging, the diagnosis of PASH on a core biopsy should not be accepted as a final diagnosis, and excisional biopsy should be performed. However, in the absence of suspicious imaging characteristics, a diagnosis of PASH at core biopsy is considered sufficient, and surgical excision is not always necessary. There is no increased risk of subsequent breast cancer associated with PASH”. The review does not mention prophylactic mastectomy as a management option.
How old do you have to be to get breast cancer?
Breast cancer is diagnosed at age 50 years or younger, with or without family history; or. 5. Women with a personal history of pancreatic adenocarcinoma at any age, or with familial pancreatic cancer, defined as having two or more first-degree relatives with pancreatic cancer.
Is a fallopian tube included in BRCA?
For the purposes of this policy, fallopian tube and primary peritoneal carcinoma should be included. Serous borderline tumor of ovary is not included. Footnotes. For purposes of this policy on BRCA testing, the term “breast cancer” includes both invasive and ductal carcinoma in situ (DCIS) breast cancers.
Is lobular carcinoma in situ included in the blood list?
Lobular carcinoma in situ (LCIS) is not included. Footnotes. Close blood relatives include first-degree relatives (e.g., mother, sister, daughter) and second-degree relatives (e.g., aunt, grandmother, niece), all of whom are on the same side of the family.
Is BRCA testing necessary for breast cancer?
BRCA testing of men with breast cancer is considered medically necessary to assess the man's risk of recurrent breast cancer and/or to assess the breast cancer risk of a female member where the affected male is a first- or second-degree blood relative of that member.

Popular Posts:
- 1. icd-10 code for bilateral aphakia
- 2. icd 10 code for chronic anxiety and depression
- 3. icd 10 code for striking against playground equipment
- 4. abnormality of gait icd-10 code number for home health
- 5. icd 10 cm code for poisoning due to inhalation of paint fumes (initial encounter)
- 6. are icd 10 code sets only required for billing to medicare and medicaid.
- 7. icd 10 code for nephrostomy tube status
- 8. icd 10 code for conducting hearing loss
- 9. 2021 icd 10 code for hypotension
- 10. icd 10 code for other weakness