Can you ever use an unspecified ICD-10 code?
In both ICD-9 and ICD-10, signs/symptoms and unspecified codes are acceptable and may even be necessary. In some cases, there may not be enough information to describe the patient's condition or no other code is available to use. Although you should report specific diagnosis codes when they are supported by the available documentation and clinical knowledge of the patient's health condition, in some cases, signs/symptoms or unspecified codes are the best choice to accurately reflect the ...
What does ICD - 10 stand for?
The International Classification of Diseases, Tenth Edition (ICD-10), is a clinical cataloging system that went into effect for the U.S. healthcare industry on Oct. 1, 2015, after a series of lengthy delays.
What is the ICD 10 code for surgical infection?
T81.49XA is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes. Short description: Infection following a procedure, other surgical site, init. The 2022 edition of ICD-10-CM T81.49XA became effective on October 1, 2021.
What is a combination code for ICD 10?
combination codes. It is the same way in ICD-10 as well, but to an entirely different level. ICD-10-CM includes hundreds of combination codes, i.e. codes that link symptoms, manifestations or complications with a particular diagnosis. The coder will have to abstract information from the medical record to comply with the specificity.

What is the CPT code for capsule endoscopy?
National Government Services, Inc. has an LCD for Colon Capsule Endoscopy (CCE) (L38571) and an LCA Billing and Coding: Colon Capsule Endoscopy (CCE) (A58294) (MCD search 02-08- 2022).
What is the meaning of capsule endoscopy?
Listen to pronunciation. (KAP-sul en-DOS-koh-pee) A procedure used to look at the inside of the intestines and other parts of the digestive tract. The patient swallows a capsule about the size of a large pill.
What are the coding guidelines for endoscopy?
The base procedure codes for GI endoscopy include 43200 (esophagoscopy), 43235 (EGD), 45330 (sig moidoscopy), and 45378 (colonoscopy) (Table 3).
Does Medicare cover capsule endoscopy?
Medicare Part B will cover non-laboratory diagnostic tests when they're medically necessary to make a diagnosis. However, it's important to remember that not all applications of capsule endoscopy may be considered medically necessary. Medicare may cover capsule endoscopy for: GI bleeding.
How many types of capsule endoscopy are there?
To date, the Given Imaging platform of capsule endoscopes includes the PillCam SB2 and SB3 for the small intestine, the PillCam ESO 2 for esophageal imaging, PillCam Colon2 for the large bowel, as well as the Agile Patency capsule (second generation) (Fig. 2.3).
What is the procedure called when you swallow a camera?
Capsule Endoscopy lets your doctor examine the lining of the middle part of your gastrointestinal tract, which includes the three portions of the small intestine (duodenum, jejunum, ileum). Your doctor will give you a pill sized video camera for you to swallow.
What is the CPT code for EGD with Bravo capsule placement?
CPT®1 code 91035, Esophagus, gastroesophageal reflux test; with mucosal attached telemetry pH electrode placement, recording, analysis and interpretation. Patient presents to the ASC for Bravo™ reflux testing system. At the encounter, a diagnostic EGD is performed for specimen or biopsy collection.
What is the ICD 9 code for endoscopy?
45.13 Other endoscopy of small intestine - ICD-9-CM Vol.
What is procedure code 43235?
Esophagogastroduodenoscopy, flexible, transoral; diagnostic, including collection of specimen(s) by brushing or washing, when performed (separate procedure)
Does insurance cover pill endoscopy?
Wireless capsule endoscopy of the esophagus will be considered medically necessary only as specified under “Covered Indications.” Performance of wireless capsule endoscopy for any other reason will be considered not medically necessary. Wireless capsule endoscopy is not reimbursable for colorectal cancer screening.
How much does it cost to have a capsule endoscopy?
Depending on where you live, capsule endoscopy can cost anywhere from $1,000 to $2,000. That still represents a savings of $750 to $1,000 compared to traditional endoscopy. Insurance pre-authorization is required for the test.
How much does PillCam cost?
The PillCam® is much more comfortable than scopes. It also makes colonoscopy possible for those who are otherwise ineligible for this important screening. Cost is another key benefit of the PillCam®. According to Slate, the camera pill price is around $500 while a colonoscopy costs an average of $4,000.
Can I sleep during capsule endoscopy?
Avoid any strenuous physical activity but make sure you keep active. Do not lie down or go to sleep. The more you move, the more active your bowel is and the capsule will progress further through your bowel. Avoid bending or stooping during the capsule endoscopy as this may cause the sensor belt to loosen.
How much is a capsule endoscopy?
Depending on where you live, capsule endoscopy can cost anywhere from $1,000 to $2,000. That still represents a savings of $750 to $1,000 compared to traditional endoscopy. Insurance pre-authorization is required for the test.
What diseases can be detected by an endoscopy?
Upper GI endoscopy can be used to identify many different diseases:gastroesophageal reflux disease.ulcers.cancer link.inflammation, or swelling.precancerous abnormalities such as Barrett's esophagus.celiac disease.strictures or narrowing of the esophagus.blockages.
What patient preparation is required for capsule endoscopy?
Drink one (1) quart of Miralax laxative the night before the procedure. Do not eat or drink after 10 p.m. on the day before the procedure. Abstain from smoking 24 hours prior to undergoing the procedure. Male patients need to shave their abdomen six inches above and below the navel on the day of the Capsule Endoscopy.
What is a capsule endoscopy?
Capsule endoscopy is an alternative to traditional endoscopy that uses a tiny camera contained within a pill that the patient swallows to obtain images of the interior of the digestive tract (for instance, to diagnose polyps or the source of bleeding).
What is the CPT code for esophagus?
For imaging of the esophagus only, using a PillCam ESO or similar Food and Drug Administration (FDA)-approved device, the correct CPT® code is 91111 Gastrointestinal tract imaging, intraluminal [e.g., capsule endoscopy], esophagus with physician interpretation and report. In this application, the camera may transmit up to 15 diagnostic images per second as it travels down the esophagus.
When is modifier 52 required for endoscopy?
For example, according to CPT®, when reporting 91110, modifier 52 Reduced services is required when the entire distance from the esophagus to the ileum is not visualized.
What is a wireless capsule?
Wireless capsules, similar (but not identical) to those used for capsule endoscopy, may be used to measure GI tract transit times and/or pressure to evaluate patients with suspected gastroparesis, slow transit constipation, unexplained diarrhea, or functional constipation that may be due to GI tract mobility issues.
How long does it take for a capsule to move through the digestive tract?
Over the course of approximately eight hours, the capsule moves through the digestive tract, transmitting pictures on average every two seconds. This type of capsule endoscopy allows for imaging of the small bowel, which is not possible with colonoscopy or EGD.
General Information
CPT codes, descriptions and other data only are copyright 2021 American Medical Association. All Rights Reserved. Applicable FARS/HHSARS apply.
CMS National Coverage Policy
Title XVIII of the Social Security Act, Section 1833 (e) states that no payment shall be made to any provider of services or other person under this part unless there has been furnished such information as may be necessary in order to determine the amounts due such provider or other person under this part for the period with respect to which the amounts are being paid or for any prior period..
Article Guidance
The billing and coding information in this article is dependent on the coverage indications, limitations and/or medical necessity described in the associated DL38837 Colon Capsule Endoscopy (CCE).
ICD-10-CM Codes that Support Medical Necessity
It is the provider’s responsibility to select codes carried out to the highest level of specificity and selected from the ICD-10-CM code book appropriate to the year in which the service is rendered for the claim (s) submitted.
ICD-10-CM Codes that DO NOT Support Medical Necessity
All those not listed under the “ICD-10 Codes that Support Medical Necessity” section of this article.
Bill Type Codes
Contractors may specify Bill Types to help providers identify those Bill Types typically used to report this service. Absence of a Bill Type does not guarantee that the article does not apply to that Bill Type.
Revenue Codes
Contractors may specify Revenue Codes to help providers identify those Revenue Codes typically used to report this service. In most instances Revenue Codes are purely advisory. Unless specified in the article, services reported under other Revenue Codes are equally subject to this coverage determination.
What is the ICd 10 code for retained foreign body fragments?
Other specified retained foreign body fragments 1 Z18.89 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes. 2 The 2021 edition of ICD-10-CM Z18.89 became effective on October 1, 2020. 3 This is the American ICD-10-CM version of Z18.89 - other international versions of ICD-10 Z18.89 may differ.
When will the ICD-10 Z18.89 be released?
The 2022 edition of ICD-10-CM Z18.89 became effective on October 1, 2021.
What is a Z00-Z99?
Categories Z00-Z99 are provided for occasions when circumstances other than a disease, injury or external cause classifiable to categories A00 -Y89 are recorded as 'diagnoses' or 'problems'. This can arise in two main ways:
What is EGD in medical terms?
For evaluation of persons with celiac disease with a positive serology who are unable to undergo esophagogastroduodenoscopy (EGD) (e.g., medically unstable, presence of known or suspected perforated viscus) with biopsy; or
Can a capsule endoscopy detect carcinoid tumors?
Capsule endoscopy has been used in detecting carcinoid tumors of the small intestine. Guidelines from the National Comprehensive Cancer Network (NCCN, 2008) recommend the use of an Octreoscan for persons who present with carcinoid syndrome to determine tumor location and extent.
Can a push enteroscopy be performed without a push endoscopy?
If capsule endoscopy is performed without a prior push enteroscopy, a push enteroscopy will still need to be performed in most cases since a negative capsule endoscopy may not exclude a lesion, and a lesion observed on capsule examination may be within reach of the enteroscope (Faigel and Fennerty, 2002).
Is capsule endoscopy only used for diagnostic purposes?
Currently, capsule endoscopy is only utilized for diagnostic purposes; individuals who require a biopsy or therapeutic intervention must then undergo a conventional endoscopic procedure. Examples of FDA approved CE devices include, but may not be limited to:
Is endoscopy contraindicated?
Capsule endoscopy is contraindicated and considered experimental and investigational under the following conditions:
Is colon capsule endoscopy effective for colorectal polyps?
Shi and colleagues (2015) stated that evaluation of mucosal inflammation is important in the management of patients with ulcerative colitis (UC). Colon capsule endoscopy has recently been shown to be effective in colorectal polyp detection. However, its role in the evaluation of mucosal inflammation in UC is unclear. This systematic review aimed to clarify the state of the art with an evidence-based summary of current studies on the utility of CCE in UC. The overall results showed that the accuracy of CCE for assessment of mucosal inflammation in UC appeared to be comparable with that of colonoscopy. Moreover, the authors stated that long-term follow-up studies with larger sample size are needed to further validate the utility of CCE in the management of UC subjects in clinical practice.
Is a wireless endoscopy necessary?
Wireless capsule endoscopy of the esophagus will be considered medically necessary only as specified under “Covered Indications.” Performance of wireless capsule endoscopy for any other reason will be considered not medically necessary.
Can WMC be administered to patients with gastric bezoar?
The WMC should not be administered to patients with a history of gastric bezoar, swallowing disorders, dysphagia, suspected strictures/ fistulae in the GI tract, physiologic gastrointestinal obstruction, GI surgery within the previous 3 months, Crohn’s disease, diverticulitis, or who have an implanted electromechanical medical device (such as pacemaker or infusion pump). The capsule is not FDA approved for use in children.
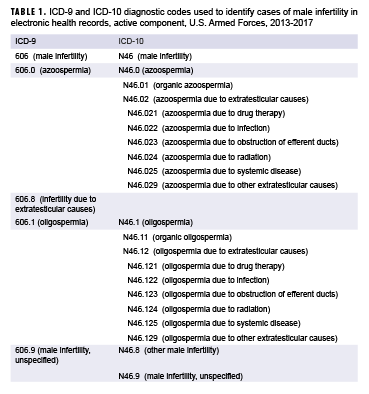
Popular Posts:
- 1. icd 10 code for suspected exposure to std
- 2. icd 10 diagnosis code for chf
- 3. icd 10 code for posterior staphyloma
- 4. icd 10 code for attention to pacemaker
- 5. icd 10 code for activity cleaning
- 6. icd 9 code for colonic ileus
- 7. icd-10-cm code for primigravida
- 8. icd 10 cm code for apositive factor ra
- 9. icd 10 code for neutropenic sepsis
- 10. icd 10 code for jaws infection