What is the ICD-10 code for Tegretol level?
What is the ICD-10 code for medication?
What is diagnosis code Z51 81?
What is the ICD-10 code for medication monitoring?
899 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes. The 2022 edition of ICD-10-CM Z79. 899 became effective on October 1, 2021. This is the American ICD-10-CM version of Z79.
What is the ICD-10 for UTI?
What is the ICD 10 code for long term use of medication?
What is Z13 89?
What is Z02 89?
What is Z76 89?
What is the ICD 10 code for high risk medication use?
Z79.
What is the ICD 10 code for diuretics?
What is the ICD 10 code for valproic acid level?
E71.
When will the ICD-10 T42.1X5A be released?
The 2022 edition of ICD-10-CM T42.1X5A became effective on October 1, 2021.
What is T42 in medicine?
T42- Poisoning by, adverse effect of and underdosing of antiepileptic, sedative- hypnotic and antiparkinsonism drugs
What is the secondary code for Chapter 20?
Use secondary code (s) from Chapter 20, External causes of morbidity, to indicate cause of injury. Codes within the T section that include the external cause do not require an additional external cause code. Type 1 Excludes.
What is a type 1 code?
Type 1 Excludes. mental or behavioral disorders due to psychoactive substance use ( F10-F19) Use Additional. code to identify the any retained foreign body, if applicable ( Z18.-) Findings of drugs and other substances, not normally found in blood.
When will ICD-10-CM R78.89 be released?
The 2022 edition of ICD-10-CM R78.89 became effective on October 1, 2021.
When will the ICd 10 Z79.2 be released?
The 2022 edition of ICD-10-CM Z79.2 became effective on October 1, 2021.
What is a Z77-Z99?
Z77-Z99 Persons with potential health hazards related to family and personal history and certain conditions influencing health status
What is the ICd 10 code for abnormal levels of drugs?
Abnormal level of other drugs, medicaments and biological substances in specimens from other organs, systems and tissues 1 R89.2 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes. 2 Short description: Abn lev drug/meds/biol subst in specimens from oth org/tiss 3 The 2021 edition of ICD-10-CM R89.2 became effective on October 1, 2020. 4 This is the American ICD-10-CM version of R89.2 - other international versions of ICD-10 R89.2 may differ.
When will ICD-10-CM R89.2 be released?
The 2022 edition of ICD-10-CM R89.2 became effective on October 1, 2021.
Does carbamazepine interfere with theophylline?
Carbamazepine may interfere with the actions of theophylline, oral contraceptives, oral anticoagulants, or doxycycline. Conversely, there have been reports indicating fluoxetine may mediate an increase in carbamazepine plasma concentrations due to inhibition of CYP3A4. 2.
Does carbamazepine affect CYP3A4?
Carbamazepine can induce the hepatic enzymes CYP3A4, CYP1A2, and CYP2C9, resulting in decreased serum levels of many drugs (eg, alprazolam, clozapine, diazepam, haloperidol, risperidone, and tricyclic antidepressants). 2.
Is carbamazepine an antiepileptic?
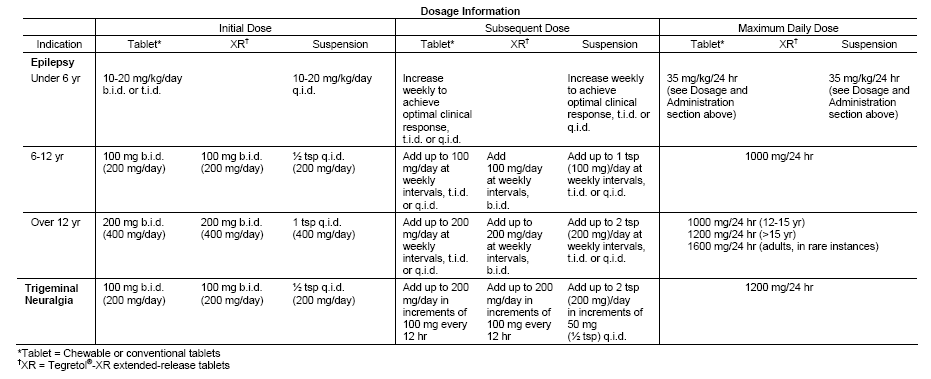
This heterocyclic (iminostilbene) compound has potent antiepileptic properties and is effective alone or with other antiepileptic drugs in partial seizures, especially complex partial seizures, generalized tonic-clonic seizures, and combinations of these seizure types. Carbamazepine generally is ineffective for absence, myoclonic, and atonic seizures. In children with symptomatic generalized epilepsy and continuous spike-and-wave discharge, these seizure types may develop or tonic-clonic seizures may increase in frequency with use of carbamazepine. It has also been noted 1 that the dose of carbamazepine cannot be used as a reliable index for predicting the serum concentration of either total or free carbamazepine serum concentrations in children with epilepsy.
Is carbamazepine safe for seizures?
Carbamazepine generally is ineffective for absence, myoclonic, and atonic seizures. In children with symptomatic generalized epilepsy and continuous spike-and-wave discharge, these seizure types may develop or tonic-clonic seizures may increase in frequency with use of carbamazepine.
Is carbamazepine better than phenobarbital?
Comparative clinical trial data indicate that patients with partial seizures may tolerate carbamazepine better than phenobarbital and primidone, but individual responses vary. Many clinicians consider carbamazepine a drug of choice for initial therapy in idiopathic and symptomatic localization-related epilepsies, especially in children and women. This drug is increasingly preferred to phenobarbital in pediatric patients because it has less effect on cognition and behavior. It is reported to have psychotropic activity that may increase alertness and elevate mood in depressed epileptic patients, but not in otherwise normal patients. Mental improvements may be due to substitution of carbamazepine for sedative drugs, control of seizures, or a direct psychotropic effect. Carbamazepine can induce the hepatic enzymes CYP3A4, CYP1A2, and CYP2C9, resulting in decreased serum levels of many drugs (eg, alprazolam, clozapine, diazepam, haloperidol, risperidone, and tricyclic antidepressants). 2
Popular Posts:
- 1. what is the icd 10 code for metabolic encephalopathy
- 2. icd 19 code for rupture membranes
- 3. icd 10 code for abscess left hand
- 4. icd 10 code for focal seizure unspecified
- 5. icd 10 code for left knee post traumatic arthritis
- 6. icd-10 code for lung cancer with mets to brain
- 7. icd 10 cm code for as
- 8. icd 10 code for pneumatocele of lung
- 9. icd 10 code for left bundle branch block on ecg
- 10. icd 10 code for left distal humerus fracture