How many codes in ICD 10?
Feb 28, 2020 · R79. 1 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes. The 2020 edition of ICD-10-CM R79. 1 became effective on October 1, 2019. Beside above, what does an elevated D dimer mean? A positive D-dimer result may indicate the presence of an abnormally high level of fibrin degradation products.
How do you code elevated D dimer?
The ICD-10-CM code R79.1 might also be used to specify conditions or terms like clotting time above reference range, coag./bleeding tests abnormal, d-dimer above reference range, deviation of international normalized ratio from target range, extrinsic coagulation pathway finding , fibrinogen screening finding, etc.
What is a valid ICD 10 code?
The ICD 10 Code for elevated liver enzymes is R94.5 and it was adopted on October 1, 2018. However, an elevated D-dimer does not always indicate the presence of a clot because a number of other factors can cause an increased level. Code: R79.89. 89. R73.0 Abnormal glucose. Wiki Elevated d-dimer. ICD-10 code R79.
What are the new ICD 10 codes?
Oct 01, 2021 · R79.1 Abnormal coagulation profile Code Version: 2022 ICD-10-CM Synonyms Clotting time above reference range Coag./bleeding tests abnormal D-dimer above reference range Deviation of international normalized ratio from target range Extrinsic coagulation pathway finding Hormone replacement therapy bleed pattern - abnormal
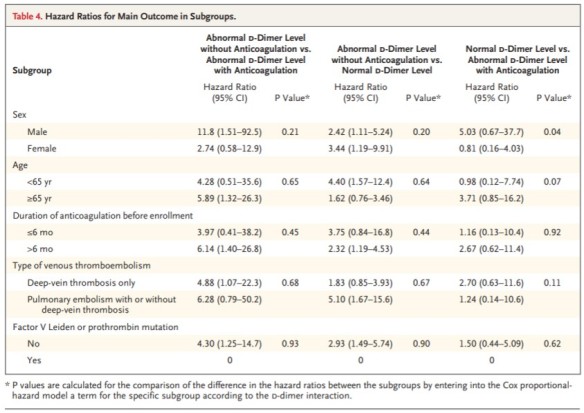
What is the ICD-10 code for an elevated D dimer?
For elevated D-dimer, look to ICD-10-CM R79. 1 Abnormal coagulation profile.Jul 28, 2017
What is R79 89 diagnosis?
Other specified abnormal findings of blood chemistryICD-10 code R79. 89 for Other specified abnormal findings of blood chemistry is a medical classification as listed by WHO under the range - Symptoms, signs and abnormal clinical and laboratory findings, not elsewhere classified .
How do you code elevated INR?
Raised INR can be coded with the ICD-10 code R79. 8 Other specified abnormal findings of blood chemistry. Z92. 1 Personal history of long-term (current) use of anticoagulants could be added if appropriate.Dec 14, 2011
What ICD-10 code covers a CMP?
Encounter for screening for other metabolic disorders The 2022 edition of ICD-10-CM Z13. 228 became effective on October 1, 2021.
What is elevated BNP ICD-10 code?
ICD-10-CM Diagnosis Code R97 R97.
What is the correct ICD-10 code for leukocytosis?
288.60 - Leukocytosis, unspecified. ICD-10-CM.
What is the ICD-10 code for subtherapeutic INR?
'Subtherapeutic INR levels' means that the patient is underwarfarinised, therefore as per ACS 0303 the correct code to assign is D68. 8 Other specified coagulation defects.Nov 6, 2020
What ICD-10 code covers PT PTT?
NCD - Partial ThromboplastinTime (PTT) (190.16)
What diagnosis covers PTT?
The PTT NCD includes in the covered list of ICD-9-CM diagnostic codes code V72. 81 (Pre-operative cardiovascular examination,) code V72. 83 (Other specified pre-operative examination,) and code V72. 84 (Pre-operative examination, unspecified).
What is diagnosis code for CBC and CMP?
General Health Panel (CPT code 80050, diagnosis code Z00. 00) – This test includes a CBC (Complete Blood Count), CMP (Comprehensive Metabolic Panel) and TSH (Thyroid Stimulating Hormone).
What ICD-10 code for CBC?
Abnormal finding of blood chemistry, unspecified The 2022 edition of ICD-10-CM R79. 9 became effective on October 1, 2021.
What ICD-10 code covers ESR?
R70.0ICD-10 code R70. 0 for Elevated erythrocyte sedimentation rate is a medical classification as listed by WHO under the range - Symptoms, signs and abnormal clinical and laboratory findings, not elsewhere classified .
Special Instructions
If the patient's hematocrit exceeds 55%, the volume of citrate in the collection tube must be adjusted. Refer to Coagulation Collection Procedures for directions.
Expected Turnaround Time
Turnaround time is defined as the usual number of days from the date of pickup of a specimen for testing to when the result is released to the ordering provider. In some cases, additional time should be allowed for additional confirmatory or additional reflex tests. Testing schedules may vary.
Collection
Blood should be collected in a blue-top tube containing 3.2% buffered sodium citrate. 1 Evacuated collection tubes must be filled to completion to ensure a proper blood to anticoagulant ratio. 2,3 The sample should be mixed immediately by gentle inversion at least six times to ensure adequate mixing of the anticoagulant with the blood.
Causes for Rejection
Gross hemolysis; clotted specimen; specimen thawed in transit; improper labeling
Use
The Innovance® D-dimer assay is intended for use in conjunction with a nonhigh clinical pretest probability (PTP) assessment model to exclude deep vein thrombosis (DVT) and pulmonary embolism (PE). 6 This test can be used to exclude VTE with nonhigh pretest probability (ie, low or low/moderate pretest probability).
Limitations
Results of this test should always be interpreted in conjunction with the patient's medical history, clinical presentation, and other findings. DVT clinical diagnosis should not be based on the result of Innovance® D-dimer alone.
Additional Information
Coagulation activation results in the cleavage of fibrinogen to fibrin monomer. 7,8 The fibrin monomers spontaneously aggregate to fibrin and are cross-linked by factor XIII; this produces a fibrin clot.
What is a blood typing test?
Blood typing is a screening test to determine blood groups and Rh antigen for blood transfusion and pregnancy. The four blood groups A, B, O, and AB are determined by the presence of antigens A and B or their absence (O) on a patient's red blood cells. In addition to ABO grouping, most immunohematology testing includes evaluation of Rh typing tests for Rh (D) antigen. Blood cells that express Rh (D) antigen are Rh positive. Red blood cells found lacking Rh (D) are considered Rh negative. Rh typing is also important during pregnancy because of the potential for mother and fetus Rh incompatiblity. If the mother is Rh negative but the father is Rh positive, the fetus may be positive for the Rh antigen. As a result, the mother’s body could develop antibodies against the Rh antigen. These antibodies may cross the placenta and cause destruction of the baby’s red blood cells, resulting in a condition known as hemolytic disease of the fetus and newborn.
Why is it important to have blood transfusions?
Transfusion of blood components of the correct blood type is necessary in order to prevent an adverse immunologic reaction. These reactions can range from very mild and sub-clinical to very severe or fatal, depending upon the components involved and condition of the recipient.

Popular Posts:
- 1. icd 10 code for squamous cell carcinoma shoulder
- 2. icd 10 cm code for nodule parotid gland
- 3. icd 10 code for mobitz type 2 av block
- 4. icd 10 code for abnormal lab results
- 5. icd 10 code for vaping use
- 6. icd 10 code for lv apical thrombus
- 7. icd 10 10 code for fungal infection
- 8. icd 10 code for acquired nasal deformity
- 9. 2017 icd 10 code for abrasion to the right heel
- 10. icd 10 cm code for nonverbal status