Hepatitis C Antibody with Reflex to HCV, RNA, Quantitative Real-Time PCR Test Code: 8472 CPT Code (s): 86803 Methodology: Immunoassay (IA) Includes: If Hepatitis C Antibody is reactive, then Hepatitis C Viral RNA, Quantitative, Real-Time PCR will be performed at an additional charge (CPT code (s): 87522).
Full Answer
What is the Order Code for HCV AB?
550354 HCV log10 log10 IU/mL 38180-6 Reflex Table for HCV Ab Order Code Order Name Result Code Result Name UofM Result LOINC Reflex 1
How many IU/ml is HCV RNA in a reflex?
HCV RNA (International Units) IU/mL 11011-4 Reflex Table for HCV Ab Order Code Order Name Result Code Result Name UofM Result LOINC Reflex 1 550362 HCV RT-PCR, Quant (Non-Graph) 550338 Hepatitis C Quantitation IU/mL 11011-4 Reflex 2 550352 HCV RNA (International Units) 550354 HCV log10 log10 IU/mL 38180-6 Reflex Table for HCV Ab Order Code
What are the results of the LOINC reflex 1 HCV AB test?
Reflex Table for HCV Ab Order Code Order Name Result Code Result Name UofM Result LOINC Reflex 1 550362 HCV RT-PCR, Quant (Non-Graph) 550314 Test Information: 19147-8 Reflex Table for HCV Ab Order Code Order Name Result Code Result Name UofM Result LOINC Reflex 1 550362 HCV RT-PCR, Quant (Non-Graph) 550396 Interpretation: 62365-2
What is the ICD 10 code for alcohol abuse and dependence?
alcohol abuse and dependence ( F10.-) chronic hepatitis NEC ( K73.-) Reimbursement claims with a date of service on or after October 1, 2015 require the use of ICD-10-CM codes.
When will the ICD-10 B18.2 be released?
The 2022 edition of ICD-10-CM B18.2 became effective on October 1, 2021.
What is Z16.-?
code to identify resistance to antimicrobial drugs ( Z16.-) Inflammation of the liver in humans that is caused by hepatitis c virus lasting six months or more. Chronic hepatitis c can lead to liver cirrhosis. 441 Disorders of liver except malignancy, cirrhosis or alcoholic hepatitis with mcc.
What are the 441 disorders of the liver?
441 Disorders of liver except malignancy, cirrhosis or alcoholic hepatitis with mcc. 442 Disorders of liver except malignancy, cirrhosis or alcoholic hepatitis with cc. 443 Disorders of liver except malignancy, cirrhosis or alcoholic hepatitis without cc/mcc. 791 Prematurity with major problems.
What is the Z22 code?
carrier or suspected carrier of infectious disease ( Z22.-) infectious and parasitic diseases complicating pregnancy, childbirth and the puerperium ( O98.-) code to identify resistance to antimicrobial drugs ( Z16.-) Inflammation of the liver in humans that is caused by hepatitis c virus lasting six months or more.
What is the code for a virus that causes a hepatitis C infection?
infectious and parasitic diseases complicating pregnancy, childbirth and the puerperium ( O98.-) code to identify resistance to antimicrobial drugs ( Z16.-) A viral infection caused by the hepatitis c virus. Inflammation of the liver in humans caused by hepatitis c virus, a single-stranded rna virus.
What is Z16 code?
code to identify resistance to antimicrobial drugs ( Z16.-) A viral infection caused by the hepatitis c virus. Inflammation of the liver in humans caused by hepatitis c virus, a single-stranded rna virus. Its incubation period is 30-90 days.
How long does it take for hepatitis C to infect the liver?
Inflammation of the liver in humans caused by hepatitis c virus, a single-stranded rna virus. Its incubation period is 30-90 days. Hepatitis c is transmitted primarily by contaminated blood parenterally, and is often associated with transfusion and intravenous drug abuse.
When will the ICD-10 B19.20 be released?
The 2022 edition of ICD-10-CM B19.20 became effective on October 1, 2021.
Is hepatitis C a viral disease?
However, in a significant number of cases, the source of hepatitis c infection is unknown. Viral disease caused by hepatitis c virus; most common form of post transfusion hepatitis, also is a common acute sporadic hepatitis; may also follow parental drug abuse.
Special Instructions
Submit two samples, preferably one refrigerated unopened gel-barrier tube and one frozen serum/plasma pour off tube.
Expected Turnaround Time
Turnaround time is defined as the usual number of days from the date of pickup of a specimen for testing to when the result is released to the ordering provider. In some cases, additional time should be allowed for additional confirmatory or additional reflex tests. Testing schedules may vary.
Container
Gel-barrier tube, lavender-top (EDTA) tube, or plasma preparation tube (PPT)
Collection
Antibody test: If tube other than a gel-barrier tube is used, transfer separated serum/plasma to a plastic transport tube.
Storage Instructions
Antibody test: refrigerated (preferred) up to 14 days. Frozen up to 14 days, room temperature up to 14 days.
Encounter For Screening For Other Viral Diseases
Z11.59 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes.
Encounter For Screening For Other Diseases And Disorders
Screening is the testing for disease or disease precursors in asymptomatic individuals so that early detection and treatment can be provided for those who test positive for the disease.
Preventive Medicine: Hepatitis C Virus Screening
Procedure Codes G0472: Hepatitis c antibody screening, for individual at high risk and other covered indication 87522: Infectious agent detection by nucleic acid hepatitis C, quantification, includes reverse transcription when performed 86804: Hepatitis C antibody confirmatory test 86803: Hepatitis C antibody
Hepatitis E Virus Screening In Peri
Sue and associates stated that autochthonous HEV infection has been reported in over 200 solid organ transplant recipients since 2006, yet little is known about the burden of HEV among SOT recipients in North America.
Encounter For Screening For Infectious And Parasitic Diseases
Screening is the testing for disease or disease precursors in asymptomatic individuals so that early detection and treatment can be provided for those who test positive for the disease.
Human Immunodeficiency Virus Testing
The overall HIV testing rate among persons with diagnoses indicating IDU during 20102016 was 8.6%, with an increase during 20102013. The rate was stable during 2014 and 2015, but it decreased during 2016. Men were less likely to have had an HIV test, compared with women .
Icdcm Diagnosis Code Z Encounter For Screening For Other Viral Diseases
Transmittal , Change Request , Dated 09/05/ for Hepatitis C Virus in Adults) Transmittal , Change Request , Dated 11/19/ for Hepatitis C Virus in Adults) Transmittal , Change Request , Dated 05/26/ ). Oct 01, · Z is a billable/specific ICDCM code that can be used to indicate a diagnosis for reimbursement purposes.
What is the CPT code for hepatitis C?
If Hepatitis C Antibody is reactive, then Hepatitis C Viral RNA, Quantitative, Real-Time PCR will be performed at an additional charge (CPT code (s): 87522) .
What is CPT code 86803?
CPT Code is subject to a Medicare Limited Coverage Policy and may require a signed ABN when ordering. CPT Code is subject to a Medicare Limited Coverage Policy and may require a signed ABN when ordering. Ordering Restrictions may apply.
Is hepatitis C a real time PCR?
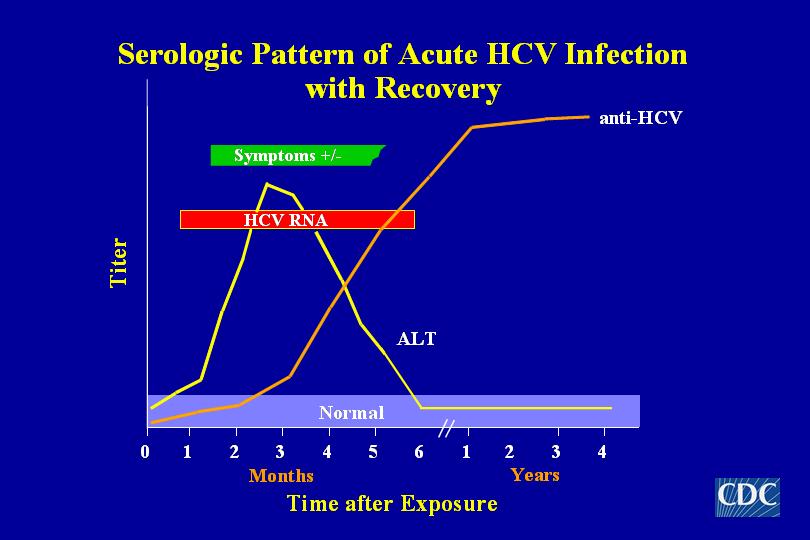
Hepatitis C Antibody with Reflex to HCV, RNA, Quantitative, Real-Time PCR - Hepatitis C Virus (HCV) is a major cause of hepatitis. The clinical symptoms of an HCV infection are variable. Infection with HCV results in a chronic infection in 50 to 80% of cases. The "window" between HCV acquisition and seroreactivity is highly variable; up to six months.
What is the CPT code for hepatitis C?
If Hepatitis C Antibody is Reactive, then Hepatitis C Viral RNA, Quantitative, Real-Time PCR will be performed at an additional charge (CPT code (s): 87522) .
What is the reflex to PCR for HCV?
Hepatitis C Antibody with Reflex to HCV RNA,PCR w/Reflex to Genotype, LiPA - Hepatitis C Virus (HCV) is the major cause of hepatitis. Making positive HCV Ab tests reflex to confirmatory molecular testing is necessary to confirm active HCV infection. Providing a second reflex to perform genotyping on HCV isolates when RNA is detected is also a necessary step before starting therapy. This double reflex will reduce the percentage of antibody-positive patients who lack appropriate confirmatory RNA testing and who do not have genotype testing performed.
What is the CPT code for Medicare?
CPT Code is subject to a Medicare Limited Coverage Policy and may require a signed ABN when ordering. Test Code. 94345. CPT Code (s) 86803. CPT Code is subject to a Medicare Limited Coverage Policy and may require a signed ABN when ordering. Ordering Restrictions may apply.
Popular Posts:
- 1. icd 9 code for history of hypercholesterolemia
- 2. icd 10 code for degenerative spondylolisthesis
- 3. icd 10 code for polyarthralgia unspecified
- 4. icd 10 code for full code status
- 5. icd 10 cm code for abdominal pain in epigastrium with hepatic cyst and calcification
- 6. icd 9 code for dominant hand monoparesis
- 7. icd-10 code for right hip pain
- 8. icd 10 code for thoracogenic scoliosis
- 9. icd 10 code for infection amputation left great toe
- 10. icd 10 code for referral to physical therapy