What is the ICD 10 code for hepatitis B?
Order Code Order Code Name Order Loinc Result Code Result Code Name UofM Result LOINC; 016881: Hep B Core Ab, IgM: 24113-3: 016881: Hep B Core Ab, IgM: 24113-3
What is the IgM test number for hepatitis B?
Oct 01, 2021 · B19.10 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes. The 2022 edition of ICD-10-CM B19.10 became effective on October 1, 2021. This is the American ICD-10-CM version of B19.10 - other international versions of ICD-10 B19.10 may differ. Applicable To Unspecified viral hepatitis B NOS
What is the ICD 10 code for hepatitis A without coma?
Oct 01, 2021 · R76.8 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes. The 2022 edition of ICD-10-CM R76.8 became effective on October 1, 2021. This is the American ICD-10-CM version of R76.8 - other international versions of ICD-10 R76.8 may differ. Applicable To.
How is hepatitis B core antigen (HBcAg) detected in the blood?
Oct 01, 2021 · B18.1 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes. The 2022 edition of ICD-10-CM B18.1 became effective on October 1, 2021. This is the American ICD-10-CM version of B18.1 - other international versions of ICD-10 B18.1 may differ. Applicable To Carrier of viral hepatitis B

What is the ICD-10 code for hepatitis screening?
Encounter for screening for other viral diseases The 2022 edition of ICD-10-CM Z11. 59 became effective on October 1, 2021.
What ICD-10 code will cover hepatitis panel?
ICD-10-CM Diagnosis Code B18 B18.
What are hepatitis B core antibodies?
Anti-HBc or HBcAb (hepatitis B core antibody) – this blood test remains positive indefinitely as a marker of past HBV infection. HBeAg (hepatitis B e-antigen) is generally detectable in patients with a new acute infection; the presence of HBeAg is associated with higher HBV DNA levels, thus, increased infectiousness.
How do you code hepatitis B?
To bill, use the ICD-9 diagnosis code to identify HIV (ICD-9 code 042) along with the appropriate CPT code (90747 for hepatitis B vaccine for immunosuppressed patient or 90632 for hepatitis A vaccine) plus the appropriate CPT adminis- tration code (90471 for immunization administration).
What is the ICD 10 code for hepatitis B?
Chronic viral hepatitis B with delta-agent B18. 0 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes. The 2022 edition of ICD-10-CM B18. 0 became effective on October 1, 2021.
What diagnosis will cover acute hepatitis panel?
The diagnosis of acute HBV infection is best established by documentation of a positive IgM antibody against the core antigen (HBcAb-IgM) and by identification of a positive hepatitis B surface antigen (HBsAg).
What is hepatitis B core IgM?
HBV has a central core and a surrounding envelope. Your immune system makes IgM antibodies for the core of HBV during the active stage of infection. Hepatitis B core IgM antibodies begin to appear in your blood several weeks after you are first infected with HBV.
What is the CPT code for hep B core Ab total?
LOINC® Codes, Performing Laboratory If Hepatitis B Core Antibody Total is reactive, then Hepatitis B Core IgM Antibody will be performed at an additional charge (CPT code(s): 86705).
What does core antibody mean?
The test for hepatitis B core antibody detects the presence of the core protein of the virus. A positive result means the person has been infected with HBV, but it does not specify whether the person has cleared the virus, still has the infection, or is immune to reinfection.
What is diagnosis code Z11 59?
52 will replace Z11. 59 (Encounter for screening for other viral diseases), which the CDC previously said should be used when patients being screened for COVID-19 have no symptoms, no known exposure to the virus, and test results that are either unknown or negative.Dec 21, 2020
What is the code 90471?
Code 90471 is used when the drug is administrated by a medical assistant or nurse and the patient does not see the physician at all. This code would also be used for any patient 19 years of age or older regardless if physician is present and does face-to-face counseling.
What is procedure code 90633?
90633 Hepatitis A vaccine, pediatric/adolescent dosage, 2 dose schedule for intramuscular use.
Special Instructions
This test may exhibit interference when sample is collected from a person who is consuming a supplement with a high dose of biotin (also termed as vitamin B7 or B8, vitamin H, or coenzyme R). It is recommended to ask all patients who may be indicated for this test about biotin supplementation.
Expected Turnaround Time
Turnaround time is defined as the usual number of days from the date of pickup of a specimen for testing to when the result is released to the ordering provider. In some cases, additional time should be allowed for additional confirmatory or additional reflex tests. Testing schedules may vary.
Collection
If tube other than a gel-barrier tube is used, transfer separated serum or plasma to a plastic transport tube.
Use
This assay can be used as an aid in the diagnosis of individuals with acute or chronic hepatitis B virus (HBV) infection and in the determination of the clinical status of HBV infected individuals in conjunction with other HBV serological markers associated with HBV infection.
Limitations
The results from this or any other diagnostic kit should be used and interpreted only in the context of the overall clinical picture. A negative test result does not exclude the possibility of exposure to hepatitis B virus. Levels of anti-HBc may be undetectable both in early infection and later after infection.
Additional Information
Hepatitis B core antigen (HBcAg), found in liver cells, does not circulate in the bloodstream. However, IgM and IgG antibodies to HBcAg can be detected serologically in HBV infected individuals. Anti-HBc IgM is detectable first and remains detectable for approximately six months.
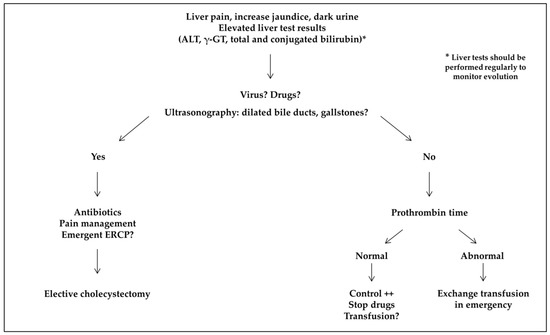
Hepatitis A,B,C,D,E and Liver Diseases
Hepatitis refer to inflammation of the liver that can caused by virus infections that affect the liver. Viral hepatitis is a relatively common disease (25 patients in every 100,000 citizens in the US) caused by a diverse group of hepatotropic agents that lead to liver inflammation and liver cell death.
CPT Codes for Hepatitis C, B Screening Test
The CDC has recommendations regarding Hepatitis screening for both hepatitis B and hepatitis C for persons at high risk for infection.
Popular Posts:
- 1. icd 10 code for stenotic cervix
- 2. icd 10 code for other congenital deformity of feet.
- 3. icd 10 code for status post aki
- 4. icd 10 code for aftercare left total hip replacement
- 5. icd 10 code for elvated bnp
- 6. icd 10 code for doe
- 7. icd 10 code for mercury screening
- 8. icd 10 code for epilepsy seizure disorder
- 9. cpt code for left foot puncture wound icd 10
- 10. icd 10 code for coughing and throat pain following inhalation of chlorine fumes from bleach