What are the new ICD 10 codes?
The new codes are for describing the infusion of tixagevimab and cilgavimab monoclonal antibody (code XW023X7), and the infusion of other new technology monoclonal antibody (code XW023Y7).
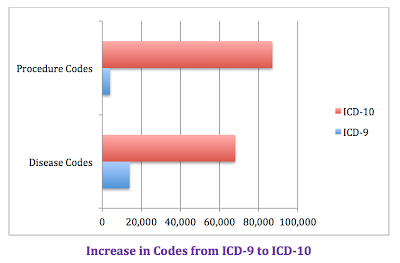
What are ICD 10 codes?
Why ICD-10 codes are important
- The ICD-10 code system offers accurate and up-to-date procedure codes to improve health care cost and ensure fair reimbursement policies. ...
- ICD-10-CM has been adopted internationally to facilitate implementation of quality health care as well as its comparison on a global scale.
- Compared to the previous version (i.e. ...
What is the ICD 10 code for history of CVA?
- Z86.73 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes.
- Short description: Prsnl hx of TIA (TIA), and cereb infrc w/o resid deficits
- The 2022 edition of ICD-10-CM Z86.73 became effective on October 1, 2021.
Where can one find ICD 10 diagnosis codes?
Search the full ICD-10 catalog by:
- Code
- Code Descriptions
- Clinical Terms or Synonyms

What ICD-10 code covers CBC with diff?
89.
What is the ICD-10 code for History of diarrhea?
OTHER COMMON GI SYMPTOM CODESColicR10.83Occult blood in feces/stoolR19.5DiarrheaR19.7Functional dyspepsia (indigestion)K30ConstipationK59.0013 more rows
What is A04 72?
72 Enterocolitis due to Clostridium difficile with toxic megacolon, without other organ complications.
What is the ICD 9 code for C. diff?
The International Classification of Diseases, 9th Revision, Clinical Modification (ICD-9) code used in this study was 008.45, "intestinal infection due to Clostridium difficile," and is the only ICD-9 code related to CDAD.
What is diagnosis code R47 89?
ICD-10 code R47. 89 for Other speech disturbances is a medical classification as listed by WHO under the range - Symptoms, signs and abnormal clinical and laboratory findings, not elsewhere classified .
Can F07 81 be used as a primary diagnosis?
Our physicians have used IDC-10 code F07. 81 as the primary diagnosis for patients presenting with post concussion syndrome.
What type of bacteria is C. diff?
C. diff is a spore-forming, Gram-positive anaerobic bacillus that produces two exotoxins: toxin A and toxin B. It is a common cause of antibiotic-associated diarrhea (AAD) and accounts for 15 to 25% of all episodes of AAD.
Is fidaxomicin a penicillin?
Fidaxomicin is in a class of medications called macrolide antibiotics. It works by killing bacteria in the intestines.
What does C. diff stand for in medical terms?
C. diff (also known as Clostridioides difficile or C. difficile) is a germ (bacterium) that causes severe diarrhea and colitis (an inflammation of the colon). It's estimated to cause almost half a million infections in the United States each year. About 1 in 6 patients who get C.
What is the ICD 9 code for diarrhea?
ICD-9 Code 787.91 -Diarrhea- Codify by AAPC.
What is the ICD-10 code for ulcerative colitis?
ICD-10 code K51 for Ulcerative colitis is a medical classification as listed by WHO under the range - Diseases of the digestive system .
What is the ICD-10 code for dementia?
90 – Unspecified Dementia without Behavioral Disturbance. ICD-Code F03. 90 is a billable ICD-10 code used for healthcare diagnosis reimbursement of Unspecified Dementia without Behavioral Disturbance.
What is the indication for Dificid?
Indication. DIFICID is a macrolide antibacterial drug indicated in adult and pediatric patients 6 months of age and older for treatment of Clostridioides difficile -associated diarrhea (CDAD).
What are the symptoms of a dificid?
Acute hypersensitivity reactions, including dyspnea, rash, pruritus, and angioedema of the mouth, throat, and face have been reported with DIFICID. If a severe hypersensitivity reaction occurs, DIFICID should be discontinued and appropriate therapy should be instituted.
What is a Dificid?
DIFICID is a macrolide antibacterial drug indicated in adult and pediatric patients 6 months of age and older for treatment of Clostridioides difficile -associated diarrhea (CDAD).
Why is Dificid used only?
To reduce the development of drug-resistant bacteria and maintain the effectiveness of DIFICID and other antibacterial drugs, DIFICID should be used only to treat infections that are proven or strongly suspected to be caused by C. difficile.
Is fidaxomicin contraindicated?
DIFICID is contraindicated in patients who have known hypersensitivity to fidaxomicin or any other ingredient in DIFICID. Acute hypersensitivity reactions, including dyspnea, rash, pruritus, and angioedema of the mouth, throat, and face have been reported with DIFICID.
Can you use Dificid for C. difficile?
Only use DIFICID for infection proven or strongly suspected to be caused by C. difficile. Prescribing DIFICID in the absence of a proven or strongly suspected C. difficile infection is unlikely to provide benefit to the patient and increases the risk of development of drug-resistant bacteria.
Popular Posts:
- 1. icd 10 code for gallbladder dysfunction
- 2. icd 10 code for uneven leg length
- 3. icd code for gallstone
- 4. icd-10 code for neurological disorder
- 5. icd 10 code for acute respiratory failure from persisten
- 6. icd 10 code for digestive disease in pregnancy
- 7. icd 10 code for hx falls
- 8. icd 10 code for associated coagulopathy
- 9. icd 10 code for degenerative disc disease at multiple levels
- 10. icd 10 dx code for cellulitis