What is the ICD 10 code for progesterone toxicity?
ICD-10-CM Diagnosis Code T38.5X5A [convert to ICD-9-CM] Adverse effect of other estrogens and progestogens, initial encounter. Adverse effect of oth estrogens and progestogens, init; Adverse effect of estrogen; Adverse effect of progesterone; Estrogen adverse reaction; Progesterone adverse reaction.
What is the ICD 10 code for fertility testing?
Encounter for fertility testing. Z31.41 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes. The 2020 edition of ICD-10-CM Z31.41 became effective on October 1, 2019. This is the American ICD-10-CM version of Z31.41 - other international versions of ICD-10 Z31.41 may differ.
What is the ICD 10 code for hormone replacement therapy?
Hormone replacement therapy. Z79.890 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes. The 2020 edition of ICD-10-CM Z79.890 became effective on October 1, 2019. This is the American ICD-10-CM version of Z79.890 - other international versions of ICD-10 Z79.890 may differ.
What is the CPT code for Nichols progesterone test?
For pre-pubertal patients, the Nichols progesterone, LC/MS/MS assay is recommended (test code 17183). Children (<18 years old): Progesterone reference ranges established on post-pubertal patient population. Reference range not established for pre-pubertal patients using this assay.
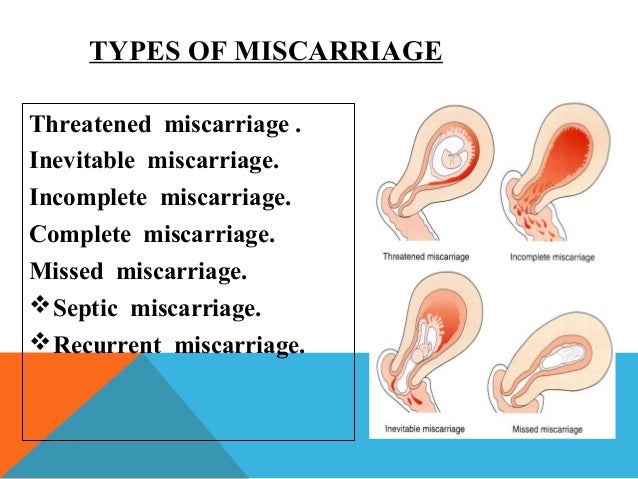
What ICD-10 code covers a progesterone?
2022 ICD-10-CM Diagnosis Code R87. 1: Abnormal level of hormones in specimens from female genital organs.
What is diagnosis code Z31 83?
Z31. 83 Encounter for assisted reprodctv fertility procedure cycle - ICD-10-CM Diagnosis Codes.
What diagnosis is Z13 29?
ICD-10 code Z13. 29 for Encounter for screening for other suspected endocrine disorder is a medical classification as listed by WHO under the range - Factors influencing health status and contact with health services .
What is diagnosis code Z31 41?
Encounter for fertility testingZ31. 41 Encounter for fertility testing - ICD-10-CM Diagnosis Codes.
What is diagnosis code Z51 11?
ICD-10 code Z51. 11 for Encounter for antineoplastic chemotherapy is a medical classification as listed by WHO under the range - Factors influencing health status and contact with health services .
What is the code for fertility testing?
"Fertility testing" - N31. 41 is the code.
What is diagnosis code Z11 3?
For claims for screening for syphilis in pregnant women at increased risk for STIs use the following ICD-10-CM diagnosis codes: • Z11. 3 - Encounter for screening for infections with a predominantly sexual mode of transmission; • and any of: Z72.
What ICD-10 code covers t3?
Abnormal results of thyroid function studies R94. 6 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes. The 2022 edition of ICD-10-CM R94. 6 became effective on October 1, 2021.
What diagnosis will cover TSH lab?
APPENDIX CDiagnoses Currently Covered by Medicare for Serum TSH TestingICD-9-CM CodePersistent (P), Thyroid (T), or Short-term (S)?Diagnosis244.0–244.9TAcquired hypothyroidism245.0–245.9TThyroiditis246.0–246.9TOther disorders of thyroid250.00–250.93PDiabetes mellitus153 more rows
What is diagnosis code E28 2?
2: Polycystic ovarian syndrome.
What does Z32 01 mean?
ICD-10 code Z32. 01 for Encounter for pregnancy test, result positive is a medical classification as listed by WHO under the range - Factors influencing health status and contact with health services .
What is the ICD-10 code for fertility preservation?
84.
What is the meaning of IVF conception?
In vitro fertilization (IVF) is a complex series of procedures used to help with fertility or prevent genetic problems and assist with the conception of a child. During IVF , mature eggs are collected (retrieved) from ovaries and fertilized by sperm in a lab.
What is the ICD-10 code for fertility preservation?
84.
What does IVF stand for in medical terms?
Listen to pronunciation. (in VEE-troh FER-tih-lih-ZAY-shun) A procedure in which eggs are removed from a woman's ovary and combined with sperm outside the body to form embryos.
What is the ICD-10 code for IVF pregnancy?
ICD-10 Code for Supervision of pregnancy resulting from assisted reproductive technology- O09. 81- Codify by AAPC.
What is the ICd 10 code for a progesterone overdose?
Poisoning by oth estrogens and progstrn, accidental, init; Estrogen overdose; Poisoning by estrogen; Poisoning by progesterone; Progesterone overdose. ICD-10-CM Diagnosis Code T38.5X1A. Poisoning by other estrogens and progestogens, ...
What is the ICd 10 code for estrogen?
ICD-10-CM Diagnosis Code T38.5X5A. Adverse effect of other estrogens and progestogens, initial encounter.
When will the ICd 10 Z79.890 be released?
The 2022 edition of ICD-10-CM Z79.890 became effective on October 1, 2021.
What is a Z77-Z99?
Z77-Z99 Persons with potential health hazards related to family and personal history and certain conditions influencing health status
What is the purpose of progesterone in fertility?
The determination of progesterone is utilized in fertility diagnosis for the detection of ovulation and assessment of the luteal phase.3,4
Where is progesterone found?
Progesterone is a steroid hormone with a molecular weight of 314.5 daltons.2Progesterone is mainly formed in the cells of the corpus luteum and during pregnancy in the placenta. Progesterone is increased in congenital adrenal hyperplasia due to 21-hydroxylase, 17-hydroxylase, and 11-β-hydroxylase deficiency. Progesterone is decreased in primary or secondary hypogonadism and short luteal phase syndrome.
What hormone is used to induce the implantation of the ovum?
Progesterone brings about the conversion of the uterine mucosa into a tissue rich in glands (secretion phase), in order to prepare for the intrauterine implantation of the fertilized ovum. During pregnancy, progesterone inhibits the contraction of the myometrium. In the mammary gland, progesterone (together with estrogens) promotes the proliferation and secretion disposition of the alveoli.2,3
Does progesterone increase during ovulation?
The progesterone concentration correlates with the development and regression of the corpus luteum. Whereas progesterone is barely detectable in the follicular phase of the female cycle, a rise in the progesterone level is observed one day prior to ovulation. Increased progesterone synthesis occurs during the luteal phase. In the second half of the cycle pregnanediol is excreted in urine as the main degradation product of progesterone.
Clinical Significance
Progesterone, Immunoassay - Levels increase sharply during the luteal phase of the menstrual cycle. The level increases from 9 to 32 weeks of pregnancy.
Test Resources
Please visit our Clinical Education Center to stay informed on any future publications, webinars, or other education opportunities.
Collection Instructions
DHEA supplementation causes a false elevation of values in this standard Immunoassay method for progesterone.
Assay Category
This test was developed and its analytical performance characteristics have been determined by Quest Diagnostics. It has not been cleared or approved by FDA. This assay has been validated pursuant to the CLIA regulations and is used for clinical purposes.
Alternative Specimen (s)
Plasma collected in: EDTA (royal blue-top) tube, sodium heparin (green-top) tube, lithium heparin (green-top) tube, or EDTA (lavender-top) tube
Collection Instructions
Collect blood in a Vacutainer® with no additives. Allow blood to clot (10-15 minutes) at room temperature. Centrifuge and separate the serum from the cells.
Setup Schedule
Collect blood in a Vacutainer® with no additives. Allow blood to clot (10-15 minutes) at room temperature. Centrifuge and separate the serum from the cells.

Popular Posts:
- 1. icd 9 code for major depression recurrent severe
- 2. icd 10 code for basal cell carcinoma of the nasal tip
- 3. icd 10 code for hematoma of pinna left
- 4. icd 10 code for left femoral neuropathy
- 5. what is the icd 10 code for non healing wound
- 6. icd 10 code for synovitis of unspecfied ankle
- 7. icd 10 code for hawking
- 8. icd 10 code for right ring finger crush injury
- 9. icd 10 code for shallow or rapid breathing
- 10. icd 10 code for dm angiopathy