Is vitamin D testing covered by Medicare?
Yes - Medicare does covers the 25 hydroxy vitamin D test. This is a screening test to detect the levels of vitamin D in the blood and it is an essential part of treatment for low bone mass and Osteoporosis. Share to failed. Install or update the app and try again. For those who can't use medicare.
What ICD 10 codes will cover vitamin D testing?
What ICD 10 codes will cover vitamin D testing?
- ICD - 10 CODE. DESCRIPTION.
- ICD - 10 CODE. DESCRIPTION.
- 2018. MEDICARE LOCAL COVERAGE DETERMINATION (LCD) - L36692.
- CPT CODES: 82306, 82652.
- Vitamin D Assay Testing.
- For services performed on or after 2-3-2017.
- DLS TEST CODE AND NAME.
What is the diagnosis code for vitamin D testing?
Starting March 1, 2022, we will only cover vitamin D screening CPT ® codes 82306, 82652 or 0038U for UnitedHealthcare commercial and Individual Exchange plan members diagnosed with one of the listed diagnosis codes in the Vitamin D Testing medical policy.
Does Medicare cover ICDs?
Part A Medicare Part A (Hospital Insurance) will cover the procedure to implant an ICD if the surgery takes place in a hospital that accepts Medicare and you have been formally admitted as an inpatient. Part A will include hospital services and supplies including general nursing, semi-private rooms, and meals.
See more
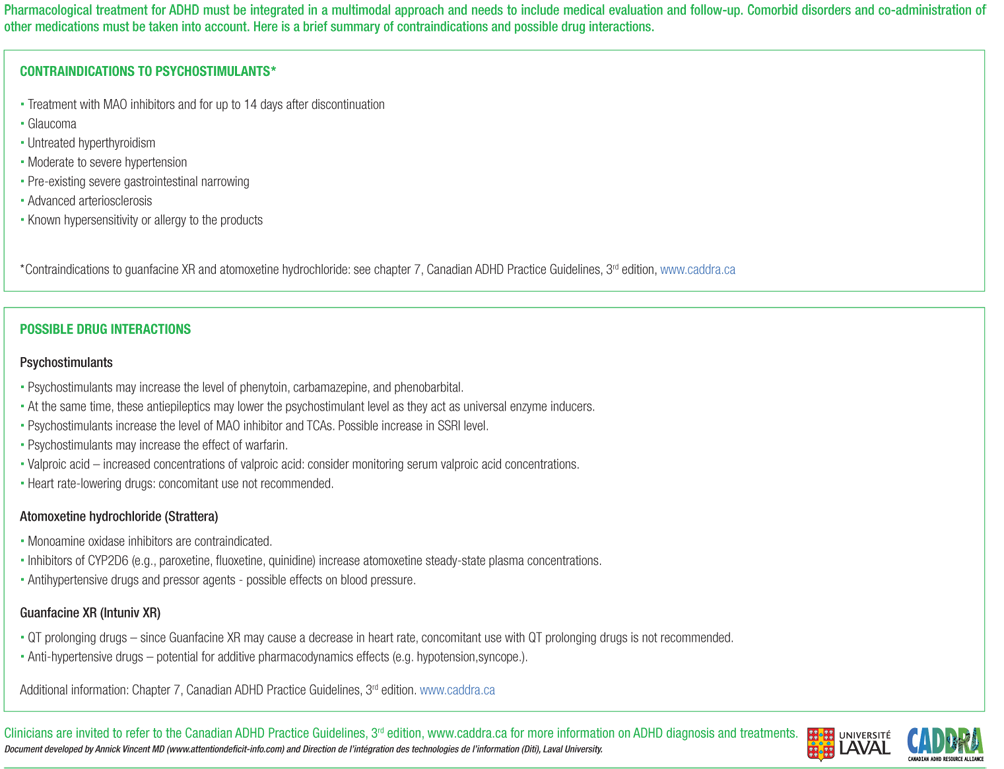
What diagnosis covers a vitamin D level?
The measurement of 25(OH) Vitamin D levels will be considered medically reasonable and necessary for patients with any of the following conditions: Chronic kidney disease stage III or greater. Hypercalcemia. Hypocalcemia.
Is vit D level covered by Medicare?
Do Medicare prescription drug plans cover vitamin D3? No. In general, Medicare prescription drug plans (Part D) do not cover this drug.
Is 82306 covered by Medicare?
CPT 82180, 82306, 82607 – Assays for Vitamins and Metabolic Function, icd CODE. Medicare generally considers vitamin assay panels (more than one vitamin assay) a screening procedure and therefore, non-covered.
What is the diagnosis code for Vitamin D 25 Hydroxy?
This test is appropriate for assessment of vitamin D deficiency. Vitamin D, 1,25-dihydroxy (CPT code 82652) is primarily indicated during patient evaluations for hypercalcemia and renal failure.
Is vitamin D blood test covered by insurance?
Reimbursement is not allowed for routine screening for vitamin D deficiency with serum testing in asymptomatic individuals and/or during general encounters. Vitamin D is an important nutrient that helps the body absorb calcium and maintain adequate bone strength.
What diagnosis code will cover 82306?
Group 1CodeDescription82306VITAMIN D; 25 HYDROXY, INCLUDES FRACTION(S), IF PERFORMED82652VITAMIN D; 1, 25 DIHYDROXY, INCLUDES FRACTION(S), IF PERFORMED
Does Medicare cover CPT code 83036?
(2) the service must be medically necessary or indicated. Once these two criteria are met, Medicare pays for most clinical laboratory tests based on the Laboratory Fee Schedule.
What CPT code covers vitamin D?
CPT 82652 Vitamin D; 1,25 Dihydroxy is determined to be medically necessary by SelectHealth commercial plans or SelectHealth Advantage only when it is ordered for patients with one of the conditions listed above.
How often can 82306 be billed?
25-OH Vitamin D-3 (82306) may be tested up to four times per year for Vitamin D deficiencies (268.0, 268.2–268.9).
What is the cost of vitamin D test?
The average cost of Vitamin D Test in India is ₹ 570 if booked through us. The average market price in India is much higher at ₹ 2800.
What blood test does Medicare pay for?
Medicare covers blood tests when they're ordered by a doctor to monitor or test for certain conditions, such as diabetes, sexually transmitted diseases, hepatitis, heart disease and other conditions. A blood test is covered by Medicare if your doctor decides it is medically necessary.
Does Medicare cover vitamin D testing for osteopenia?
For Medicare beneficiaries, screening tests are governed by statute. Vitamin D testing may not be used for routine screening. Once a beneficiary has been shown to be vitamin D deficient, further testing is medically necessary only to ensure adequate replacement has been accomplished.
Does Medicare pay for vitamin B12 blood test?
The Centers for Medicare & Medicaid Services also do not provide coverage for routine testing for vitamin B12 deficiency. There is agreement within the literature that serum vitamin B12 testing should be used to diagnose vitamin B12 deficiency in symptomatic and high-risk populations.
What is the optimal clinical indicator of vitamin D metabolism?
II. Serum concentration of 25 hydroxyvitamin D (25OHD) is the optimal clinical indicator of vitamin D metabolism due to the rapid conversion of vitamin D to 25 OHD with only a small fraction converted to 1,25 hydroxyvitamin D (1, 25 OHD).
Why is vitamin D called a vitamin?
Vitamin D is called a “vitamin” because of its exogenous source, predominately from oily fish in the form of vitamin D2 and vitamin D3.
What is the Task Force on Vitamin D?
The Endocrine Society Task Force for Evaluation, Treatment and Prevention of Vitamin D deficiency (2011) recommended screening for vitamin D deficiency in individuals at risk for deficiency. The Task Force did not recommend population screening for vitamin D deficiency in individuals who are not at risk (high quality evidence).
How long does vitamin D last in the body?
A major source of vitamin D for most humans comes from exposure of the skin to sunlight typically between 1000 hours and 1500 hours in the spring, summer, and fall. Vitamin D produced in the skin may last at least twice as long in the blood compared with ingested vitamin D.
How much vitamin D3 should I take daily?
The USPSTF recommends against daily supplementation with 400 IU or less of vitamin D3 and 1,000 mg or less of calcium for the primary prevention of fractures in non-institutionalized postmenopausal women. (Grade: D Recommendation).
What happens if you don't take vitamin D?
Vitamin D deficiency results in abnormalities in calcium, phosphorus, and bone metabolism. Specifically, vitamin D deficiency causes a decrease in the efficiency of intestinal calcium and phosphorus absorption of dietary calcium and phosphorus, resulting in an increase in parathyroid hormone (PTH) levels.
What is the most infamous vitamin D deficiency?
Vitamin D deficiency may lead to a variety of disorders, the most infamous of which is rickets. Evaluating patients’ vitamin D levels is accomplished by measuring the level of 25-hydroxyvitamin D. Measurement of other metabolites is generally not medically necessary.
What is the ICd 10 code for vitamin D deficiency?
E55.9* If more than one LCD-listed condition contributes to vitamin D deficiency in a given patient and/or is improved by vitamin D administration, coders should use: ICD-10 E55.9 UNSPECIFIED VITAMIN D DEFICIENCY. This code should not be used for any other indication.
What is the recommended level of vitamin D for repeat testing?
If Vitamin D level is between 20 and 50 ng/ml and patient is clinically stable, documentation in the patient's medical record for repeat testing must clearly indicate the necessity of the test.
Can you use CPT in Medicare?
You, your employees and agents are authorized to use CPT only as contained in the following authorized materials of CMS internally within your organization within the United States for the sole use by yourself, employees and agents. Use is limited to use in Medicare, Medicaid or other programs administered by the Centers for Medicare and Medicaid Services (CMS). You agree to take all necessary steps to insure that your employees and agents abide by the terms of this agreement.
Does CMS have a CDT license?
Organizations who contract with CMS acknowledge that they may have a commercial CDT license with the ADA, and that use of CDT codes as permitted herein for the administration of CMS programs does not extend to any other programs or services the organization may administer and royalties dues for the use of the CDT codes are governed by their commercial license.
Can you use CPT in Medicare?
You, your employees and agents are authorized to use CPT only as contained in the following authorized materials of CMS internally within your organization within the United States for the sole use by yourself, employees and agents. Use is limited to use in Medicare, Medicaid or other programs administered by the Centers for Medicare and Medicaid Services (CMS). You agree to take all necessary steps to insure that your employees and agents abide by the terms of this agreement.
Does CMS have a CDT license?
Organizations who contract with CMS acknowledge that they may have a commercial CDT license with the ADA, and that use of CDT codes as permitted herein for the administration of CMS programs does not extend to any other programs or services the organization may administer and royalties dues for the use of the CDT codes are governed by their commercial license.
What is the revision of ICD-10?
Explanation of Revision: This LCD was revised in the “ICD-10 Codes that Support Medical Necessity” section of the LCD under “Group 1 Medical Necessity ICD-10 Codes Asterisk Explanation:” to include an explanation that all the codes within the asterisked range from the first code to the last code apply. The effective date of this revision is based on process date.
How many Vitamin D tests are done per year?
Patients with Vitamin D deficiency that have been supplemented to normal levels are limited to one test per year.
When was the LCD revised?
LCD revised and published on 10/29/2020. Title XVIII of the Social Security Act, Section 1833 (e) reference, CPT codes 82306 and 82652 removed from LCD to comply with CR 10901, also LCD was updated to be consistent with GWS template.
Can you use CPT in Medicare?
You, your employees and agents are authorized to use CPT only as contained in the following authorized materials of CMS internally within your organization within the United States for the sole use by yourself, employees and agents. Use is limited to use in Medicare, Medicaid or other programs administered by the Centers for Medicare and Medicaid Services (CMS). You agree to take all necessary steps to insure that your employees and agents abide by the terms of this agreement.
Does CMS have a CDT license?
Organizations who contract with CMS acknowledge that they may have a commercial CDT license with the ADA, and that use of CDT codes as permitted herein for the administration of CMS programs does not extend to any other programs or services the organization may administer and royalties dues for the use of the CDT codes are governed by their commercial license.
What is the correct assay for vitamin D?
Once a beneficiary has been shown to be Vitamin D deficient, by assay or clinical findings, the correctly chosen assay (25 hydroxyvitamin D, or 1,25 di-hydroxyvitamin D) may be used to assure correct supplementation to attain the serum levels outlined in Limitations. Continued findings outside those parameters (again outlined in the Limitations section) may warrant additional testing.
What is the pragmatic approach to vitamin D?
A pragmatic approach for patients and their physicians was developed by the ABIM Foundation in its Choosing Wisely initiative. The patient friendly literature reassures individuals that healthy diet and exercise maintain most persons in an adequate range of Vitamin D level. It raises the possible justification of empiric vitamin D supplementation without testing for those patients without risk factors but may be thought to have inadequate sun exposure or dietary intake, while outlining those clinical risk factors that warrant baseline diagnostic assays.
Why is 25 hydroxyvitamin D needed?
In its 2011 report, the Institute of Medicine shared the concerns that a “reassessment of laboratory ranges for 25-hydroxyvitamin D” was needed to decrease risks of over and under treatment of Vitamin D deficiency.
How many IU of cholecalciferol is needed for hip fracture?
A second meta-analysis pooled 12 randomized controlled trials, all using cholecalciferol supplementation therapy between 700-800 IU/d. The results demonstrated a reduction in fractures of the hip of 26%, and non-vertebral fractures of 23%, in both ambulatory and institutionalized elderly persons.
Is vitamin D good for health?
The benefits of treatment of Vitamin D supplementation may be modest, and those benefits made difficult to quantify by general health, habit s such as exercise and smoking, and other contributory factors such as ethnicity and medication treatment regimens.
Can you use CPT in Medicare?
You, your employees and agents are authorized to use CPT only as contained in the following authorized materials of CMS internally within your organization within the United States for the sole use by yourself, employees and agents. Use is limited to use in Medicare, Medicaid or other programs administered by the Centers for Medicare and Medicaid Services (CMS). You agree to take all necessary steps to insure that your employees and agents abide by the terms of this agreement.
Is 25 hydroxyvitamin D a metabolite?
It is established that 25-hydroxyvitamin D is more reflective of total body stores of vitamin D than the shorter lived, active metabolite, 1,25 dihydroxyvitamin D. Although lack of laboratory standardization is commonly noted in most papers, it is the preferred initial assay in the evaluation of most patients with hypovitaminosis D. The 25-hydroxyvitamin D undergoes additional hydroxylation in the kidney by 1- alphahydroxylase under the influence of parathyroid hormone to produce the active metabolite. The 1,25 dihydroxyvitamin D assay is reserved for those patients where a contributory medical illness generally related to kidney disease, but also possibly related to liver, parathyroid or genetic diseases that may influence this normal metabolism.
Popular Posts:
- 1. 2016 icd 10 code for lung cancer
- 2. icd 10 code for lesion shoulder
- 3. icd 9 code for facial palsy
- 4. icd 10 code for pneumomediastinum
- 5. icd 10 code for memory hs
- 6. what is the icd 10 code for riyria
- 7. icd 10 code for blunt head trauma
- 8. icd 10 code for curvature of spine
- 9. icd 9 diagnosis code for reversible inferior ischemia
- 10. icd 10 code for postop wound bleeding