What is the ICD 10 code for heart valve replacement?
Presence of other heart-valve replacement. 2016 2017 2018 2019 Billable/Specific Code POA Exempt. Z95.4 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes. The 2018/2019 edition of ICD-10-CM Z95.4 became effective on October 1, 2018.
Can a C code be used for heart valve surgery?
Notes: Surgical heart valve procedures are not allowed in Outpatient or Non-Facility settings. Level II HCPCS codes, including C-codes, are not applicable to surgical valve procedures. C-codes are used in conjucntion with the Medicare prospective payment system for outpatient procedures only.
What is the CPT code for replacement of tricuspid valve?
Replacement of Tricuspid Valve with Autologous Tissue Substitute, Open Approach 02RJ08Z Replacement of Tricuspid Valve with Zooplastic Tissue, Open Approach ... CPT Code Procedure Description ICD-10 PCS Code Descruption Central ECMO Extracorporeal Oxygenation, Membrane, Central 5A1522F Veno-arterial
What is the ICD 10 code for implantable heart surgery?
Diagnosis Index entries containing back-references to Z95.2: Presence (of) artificial heart (fully implantable) (mechanical) Z95.812 ICD-10-CM Diagnosis Code Z95.812 Replacement by artificial or mechanical device or prosthesis of heart Z95.812 ICD-10-CM Diagnosis Code Z95.812
What is the ICD 10 code for mechanical heart valve?
Breakdown (mechanical) of heart valve prosthesis, initial encounter. T82. 01XA is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes. The 2022 edition of ICD-10-CM T82.
Is St Jude valve a mechanical valve?
Two of the most widely used mechanical heart valves are the standard St. Jude Medical® Mechanical Heart Valve (SJM; St. Jude Medical Inc.; Minneapolis, Minn) and the CarboMedics Standard Aortic Valve (CM; CarboMedics, Inc.; Austin, Tex) bileaflet valve prostheses.
Is St Jude valve mechanical or bioprosthetic?
Background: The St Jude Medical Epic heart valve (St Jude Medical, Inc, St Paul, Minn) is a tricomposite glutaraldehyde-preserved porcine bioprosthesis.
Which type of valve is a St Jude prosthesis?
The St. Jude Medical cardiac valve is a low-profile, bileaflet, central-flow prosthesis made entirely of pyrolytic carbon. A total of 261 St. Jude valves were implanted in 253 patients during a 25-month period.
What is the difference between bioprosthetic valve and mechanical valve?
Artificial heart valves are often known as mechanical heart valves and made from metallic alloys or plastic materials. In bioprosthetic heart valves, the valve tissue is typically from an animal species and mounted on a frame, known as a bioprosthesis.
What is the best mechanical aortic valve?
The On-X Aortic Valve, as a mechanical heart valve, has a much lower risk of reoperation than tissue (bioprosthetic) valves, with the additional benefit of less bleeding risk than other mechanical aortic valves because of the significantly lower amount of blood thinner required.
Is a mechanical valve a prosthetic valve?
A mechanical prosthesis is recommended for aortic valve replacement (AVR) in patients with a mechanical valve in the mitral or tricuspid position (class I). A bioprosthesis is recommended for AVR in patients of any age who will not take warfarin or who have major medical contraindications to warfarin therapy (class I).
Is a mechanical heart valve a prosthetic?
The most common mechanical valve is the bileaflet valve introduced in 1977 [15]. Mechanical valve prostheses are usually recommended for patients aged under 60 years, because these prostheses are durable with the potential to last over 20 years and often do not require replacement surgeries [16–18].
What is a bioprosthetic heart valve?
Bioprosthetic valves are generally made of either bovine pericardium or porcine aortic valves, but may also be produced from equine or porcine pericardium. The advantage of these bioprosthetic valves is that they do not require life-long anticoagulation.
What kind of valve is the aortic valve?
semilunar valvesThe normal human heart contains 4 valves that regulate blood flow into and out of the heart. The aortic and pulmonic valves are known as the semilunar valves, whereas the tricuspid and mitral valves are referred to as the atrioventricular valves.
Which anticoagulant is contraindicated with mechanical heart valve?
Dabigatran is contraindicated — Dabigatran is contraindicated in patients with mechanical heart valves.
What is a tissue valve?
Tissue Valve Tissue valves are created from animal donors' valves or animal tissue that's strong and flexible. Tissue valves can last 10 to 20 years, and usually don't require the long-term use of medication.
How long can you live with mechanical heart valve?
If your heart was already working poorly before your valve was replaced, you may still have heart problems. Mechanical valves don't usually wear out. They usually last 20 years or more. Other problems might happen with the valve, such as an infection.
What kind of valve is the aortic valve?
semilunar valvesThe normal human heart contains 4 valves that regulate blood flow into and out of the heart. The aortic and pulmonic valves are known as the semilunar valves, whereas the tricuspid and mitral valves are referred to as the atrioventricular valves.
Can mechanical heart valves be replaced?
Mechanical valve replacement To replace a heart valve, your doctor removes the heart valve and replaces it with a mechanical valve or a valve made from cow, pig or human heart tissue (biological tissue valve). Biological valves often eventually need to be replaced, as they break down over time.
What is the life expectancy of someone with an artificial heart valve?
Median survival time was 10.9 years (95% confidence interval: 10.6-11.2 years) in low-risk, 7.3 years (7.0-7.9 years) in intermediate-risk, and 5.8 years (5.4-6.5 years) in high-risk patients.
What is the ICD-10 code for a procedure?
Physicians use ICD-10 CM codes for diagnoses and CPT codes for procedures, regardless of whether the setting is inpatient or outpatient. The ICD-10 CM diagnosis codes are used for claims adjudication. However, for determining Medicare payment, only the CPT procedure codes are used. For Medicare, physician reimbursement is under the RBRVS system. Each CPT code is assigned a unique relative value unit, which is then converted into the payment amount. Medicare has used RBRVS for physician reimbursement since 1992.
What is the ICD-10 code for inpatient admission?
Hospitals assign ICD-10 codes for both diagnoses and procedures for inpatient admissions. For Medicare, inpatient hospital reimbursement is under the Medicare Severity Diagnosis Related Groups (MS-DRG) system. For each admission, the ICD-10 diagnosis and procedure codes are grouped into one of over 750 MS-DRGs. Regardless of the number of codes, only one MS-DRG is assigned to the admission. Each MS-DRG has a unique relative weight, which is then converted into the payment amount. Medicare has used the DRG system for hospital inpatient reimbursement since 1983.
What is a C code?
C codes do not apply to inpatient surgical procedures such as CABG or valve replacement procedures. C codes are used in conjunction with the Medicare prospective payment system for outpatient procedures (APCs).
What is the procedure code for mitral valve replacement?
The procedure code 02RG3JZ is in the medical and surgical section and is part of the heart and great vessels body system, classified under the replacement operation. The applicable bodypart is mitral valve.
What is ICD-10-PCS?
The ICD-10 Procedure Coding System (ICD-10-PCS) is a catalog of procedural codes used by medical professionals for hospital inpatient healthcare settings. The Centers for Medicare and Medicaid Services (CMS) maintain the catalog in the U.S. releasing yearly updates. These 2022 ICD-10-PCS codes are to be used for discharges occurring from October 1, 2021 through September 30, 2022.
How many decimals are in the ICD-10 code?
Each ICD-10-PCS code has a structure of seven alphanumeric characters and contains no decimals . The first character defines the major "section". Depending on the "section" the second through seventh characters mean different things.
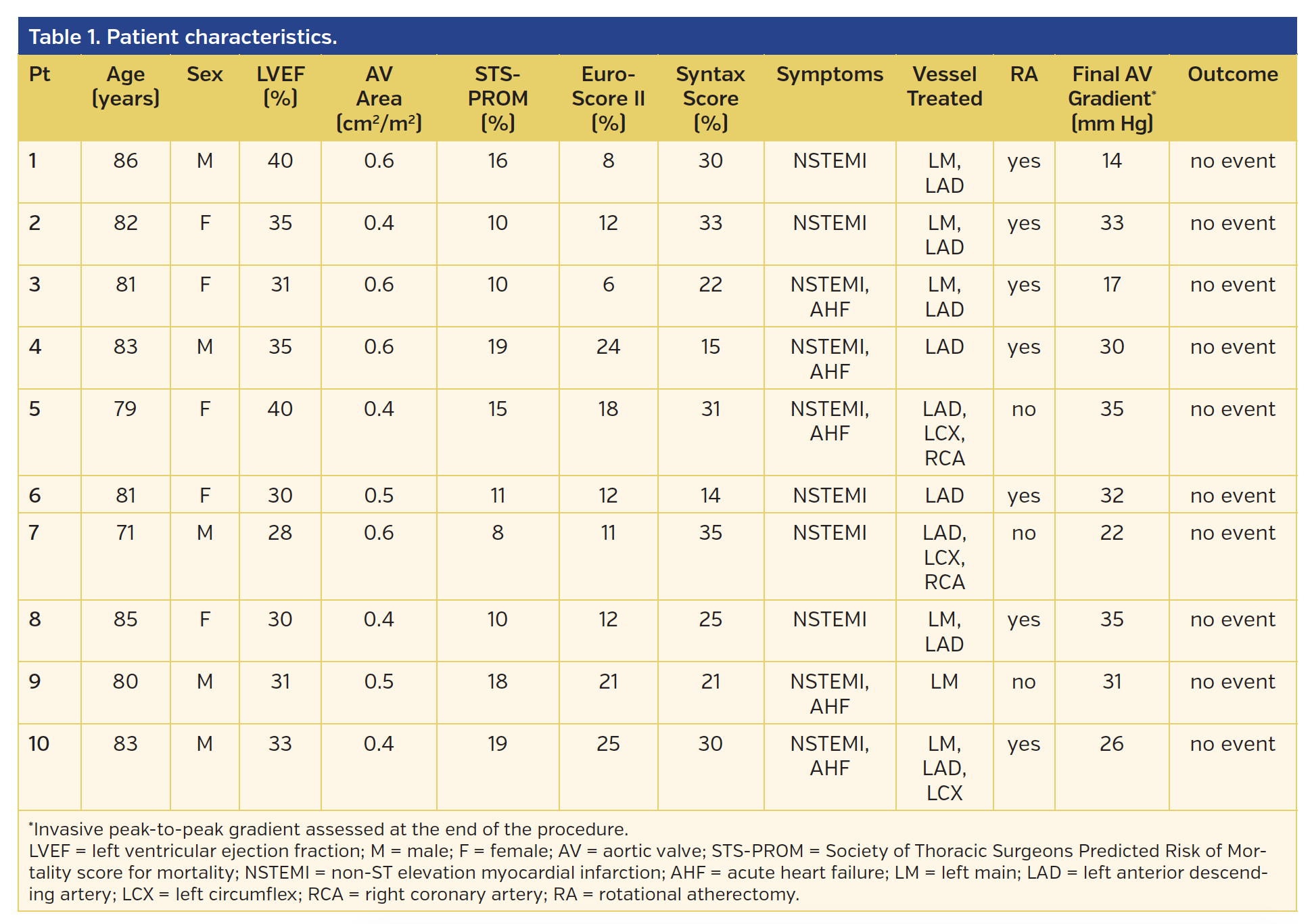
What is the SJM valve made of?
This is one of the early versions of the St. Jude Medical (SJM) artificial heart valve, a bi-leaflet valve made of pyrolytic carbon. There are four pivot points, and the edges of the donut are not as rounded as later models. A white Dacron sewing ring surrounds the valve.
Who invented the SJM valve?
The SJM valve quickly became the 'gold standard' for subsequent valves. St. Jude Medical was founded by Manuel "Manny" Villafaña in 1976, in St. Paul, Minnesota, U.S.A. The model for its popular bi-leaflet valve was developed at the University of Minnesota in 1972 by Dr. Demetre Nicoloff (1934-2003). It differed from previous valves ...
When was the first SJM valve implant made?
The first surgical implant with a SJM heart valve was carried out in October 1977. The SJM valve was the first all carbon valve in clinical use. All St. Jude valves would be made with pyrolytic carbon, a material and coating recognized for its biocompatibility and thromboresistance.
Popular Posts:
- 1. icd 9 code for 300.01
- 2. icd 10 code for anca associated vasculitis
- 3. icd 9 code for muscle aches
- 4. icd 10 code for the blood bank technician prepares 6 units of blood for freezing
- 5. icd 10 code for right hydrocele sac
- 6. icd code for internal bleeding
- 7. icd-10-cm diagnosis code for fever
- 8. icd 10 code for bilateral maxillary mucosal thickening
- 9. icd 10 code for acute excerbation of chronic sinusitis
- 10. icd 10 code for dietary weight loss