What is the ICD 9 code for anticoagulant?
2012 ICD-9-CM Diagnosis Code V58.61. Long-term (current) use of anticoagulants. Short description: Long-term use anticoagul. ICD-9-CM V58.61 is a billable medical code that can be used to indicate a diagnosis on a reimbursement claim, however, V58.61 should only be used for claims with a date of service on or before September 30, 2015.
What is the D68 code for anticoagulant?
D68 codes are diagnosis code for a diagnosis of a coagulation defect that must be rendered by the provide. To be on an anticoagulant doe not mean the patient has a coagulation defect. When a patient is on a drug, any drug, and the are there for monitoring purposes then you use Z51.81 for drug monitoring and the Z79 code for the drug.
What is the ICD 9 cm code for diagnosis?
ICD-9-CM V58.61 is a billable medical code that can be used to indicate a diagnosis on a reimbursement claim, however, V58.61 should only be used for claims with a date of service on or before September 30, 2015. For claims with a date of service on or after October 1, 2015, use an equivalent ICD-10-CM code (or codes).
How is anticoagulation monitored in patients on warfarin?
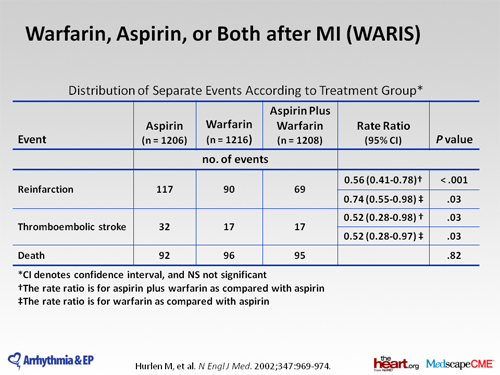
Patient self-testing/self management through the use of a home prothrombin monitor is another method of monitoring anticoagulation and presently represents < 5% of patients being anticoagulated. In general, most patients who are stable on chronic warfarin therapy are tested approximately once a month.
What is the ICD 10 code for anticoagulated?
ICD-10 code Z79. 01 for Long term (current) use of anticoagulants is a medical classification as listed by WHO under the range - Factors influencing health status and contact with health services .
What is the diagnosis code for Coumadin therapy?
Long term (current) use of anticoagulants Z79. 01 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes. The 2022 edition of ICD-10-CM Z79. 01 became effective on October 1, 2021.
How do you code long term use of anticoagulants?
01 Long term (current) use of anticoagulants.
What is the ICD 10 code for long term use of anticoagulants?
01 - Long term (current) use of anticoagulants.
What diagnosis code will cover PT INR?
1: Abnormal coagulation profile.
What diagnosis will cover CPT 85610?
A: When physicians use a prothrombin time test (reported with CPT code 85610) to monitor patients on anticoagulant drugs, Medicare pays the entity that performed the test. Its payment for the test is based on the geographically specific laboratory test fee schedule.
Is Coumadin an anticoagulant?
Warfarin (brand name: Jantoven) is a prescription medication that interferes with normal blood clotting (coagulation). It is also called an anticoagulant. Warfarin is sold under the brand name Coumadin in some countries; however, this brand is no longer available in the United States or Canada.
What is Chronic anticoagulation?
Long-term anticoagulation is necessary to prevent the high frequency of recurrent venous thrombosis or thromboembolic events. Interruption of anticoagulation within the first 12 weeks of therapy appears to result in a 25% incidence of recurrent thrombosis.
How do you bill for anticoagulation management?
CPT code 99211 is the lowest level evaluation and management (E/M) service and does not require a physician face-to-face encounter with the patient.
What is the ICD-10 code for long-term use of medication?
The ICD-10 section that covers long-term drug therapy is Z79, with many subsections and specific diagnosis codes.
What is diagnosis code z01818?
Encounter for other preprocedural examination818, “Encounter for other preprocedural examination.” Most pre-op exams will be coded with Z01. 818. The ICD-10 instructions say to use the preprocedural diagnosis code first, and then the reason for the surgery and any additional findings.
What ICD-10 code covers PT PTT?
NCD - Partial ThromboplastinTime (PTT) (190.16)
What are ICD-10 codes physical therapy?
Common ICD-10 Codes for Physical TherapyM25.50. Pain in unspecified joint.M25.511. Pain in right shoulder.M25.512. Pain in left shoulder.M25.519. Pain in unspecified shoulder.M25.521. Pain in right elbow.M25.522. Pain in left elbow.M25.529. Pain in unspecified elbow.M25.531. Pain in right wrist.More items...
Are there ICD-10 procedure codes?
ICD-10-PCS will be the official system of assigning codes to procedures associated with hospital utilization in the United States. ICD-10-PCS codes will support data collection, payment and electronic health records. ICD-10-PCS is a medical classification coding system for procedural codes.
What ICD-10 code covers PT PTT?
NCD - Partial ThromboplastinTime (PTT) (190.16)
What are ICD-10 codes used for?
The ICD-10-CM (International Classification of Diseases, Tenth Revision, Clinical Modification) is a system used by physicians and other healthcare providers to classify and code all diagnoses, symptoms and procedures recorded in conjunction with hospital care in the United States.
How long do you have to be anticoagulated before using an INR?
The patient must have been anticoagulated for at least three months prior to use of the home INR device; and
What is the NCD number for prothrombin time?
This NCD is distinct from and makes no changes to the Prothrombin Time clinical laboratory NCD at 190.17 of the National Coverage Determinations Manual.
What is the TTR in anticoagulation?
Unless citing the work of others, we use the term "TTR" in this memorandum to refer to time in therapeutic target range. This is defined as the number of patient-days of follow-up which were within target range divided by the total number of patient-days included in the follow-up period (Samsa and Matchar 1999). The scope of this memorandum is not limited by the use of alternative nomenclature.
How many tests are approved for prothrombin?
FDA has cleared 6 tests for prescriptive home use for prothrombin assays, only three are active at this point.
Does Medicare cover prothrombin?
Our current National Coverage Determination (NCD) is at § 190.11 of the Medicare NCD manual, coverage is limited to patients with mechanical heart valves. After examining additional medical evidence, we are expanding Medicare coverage of home prothrombin (INR) monitoring to include chronic atrial fibrillation and venous thromboembolism under the following conditions:
Did CMS convene the Evidence Development and Coverage Advisory Committee for this analysis?
CMS did not convene the Medicare Evidence Development and Coverage Advisory Committee for this analysis.
Can warfarin cause bleeding?
Warfarin sodium can cause major or fatal bleeding. Bleeding is more likely to occur during the starting period and with a higher dose (resulting in a higher INR). Risk factors for bleeding include high intensity of anticoagulation (INR >4.0), age ≥65, highly variable INRs, history of gastrointestinal bleeding, hypertension, cerebrovascular disease, serious heart disease, anemia, malignancy, trauma, renal insufficiency, concomitant drugs (see PRECAUTIONS) and long duration of warfarin therapy. Regular monitoring of INR should be performed on all treated patients. Those at high risk of bleeding may benefit from more frequent INR monitoring, careful dose adjustment to desired INR, and a shorter duration of therapy. Patients should be instructed about prevention measures to minimize risk of bleeding and to report immediately to physicians signs and symptoms of bleeding (see PRECAUTIONS: Information for Patients ).
When will the ICd 10 Z79.01 be released?
The 2022 edition of ICD-10-CM Z79.01 became effective on October 1, 2021.
What is a Z77-Z99?
Z77-Z99 Persons with potential health hazards related to family and personal history and certain conditions influencing health status
What is the difference between Z79 and D58?
The difference between the D58 and the Z79 codes is the D68 must be a rendered diagnosis and the z79 is a code for the status of being on a medication. use the Z51.81 for the drug monitoring followed by the Z79.01 for the anticoagulant followed by the Z code for the history of the DVT.
What does Z51.81 mean?
To be on an anticoagulant doe not mean the patient has a coagulation defect. When a patient is on a drug, any drug, and the are there for monitoring purposes then you use Z51.81 for drug monitoring and the Z79 code for the drug.
Can you use D68 code for coagulation defect?
mitchellde. Yes the provider will need to render a diagnosis of a coagulation defect before you could use the D68 code. The PE would be code the same as any other anticoag check encounter the Z51.81 the Z79.01 and the history of the PE.
Popular Posts:
- 1. icd-10-cm for hospitals: the complete official code set 2016
- 2. icd 10 code for congestive heart
- 3. id-10 code for \\\\\\\\\\\\\\\\\\\\\\\\\\icd-10 code for laceration right forearm
- 4. icd 10 code for chf exacerbation
- 5. icd 10 cm code for head injury.
- 6. icd 10 code for major depressive disorder unspecified
- 7. icd 10 code for diabetes mellitus type 2 uncomplicated controlled by diet
- 8. icd-10 code for dermoid cyst
- 9. icd 10 code for psychophysiological insomnia
- 10. icd 10 code for bed bug bites