How long can you live with an implanted defibrillator (ICD)?
ICD-9 Code V45.02 Automatic implantable cardiac defibrillator in situ. ICD-9 Index; Chapter: E; Section: V40-V49; Block: V45 Other postsurgical states; V45.02 - Status autm crd dfbrltr
What is the best portable defibrillator?
defibrillator, automatic implantable cardiac (with synchronous cardiac pacemaker) V45.02 V45.01 ICD9Data.com V45.09 ICD-9-CM codes are used in medical billing and coding to describe diseases, injuries, symptoms and conditions. ICD-9-CM V45.02 is one of thousands of ICD-9-CM codes used in healthcare.
What is the recovery time for a defibrillator implant?
Billable Medical Code for Automatic Implantable Cardiac Defibrillator in Situ Diagnosis Code for Reimbursement Claim: ICD-9-CM V45.02. Code will be replaced by October 2015 and relabeled as ICD-10-CM V45.02. The Short Description Is: Status autm crd dfbrltr. Known As
What is the difference between a pacemaker and an ICD?
Automatic implantable cardiac defibrillator in situ (V45.02) ICD-9 code V45.02 for Automatic implantable cardiac defibrillator in situ is a medical classification as listed by WHO under the range -PERSONS WITH A CONDITION INFLUENCING THEIR HEALTH STATUS (V40-V49). Subscribe to Codify and get the code details in a flash.

What is the ICD-10 code for defibrillator?
Z95. 810 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes. The 2022 edition of ICD-10-CM Z95. 810 became effective on October 1, 2021.
What ICD-9 codes?
Is ICD-9 still used in 2020?
Currently, the U.S. is the only industrialized nation still utilizing ICD-9-CM codes for morbidity data, though we have already transitioned to ICD-10 for mortality.
Are we using ICD-9 or 10?
How can you tell the difference between ICD-9 and ICD-10 codes?
What is ICD-9 and CPT coding?
What are the new 2020 ICD-10 codes?
- Eye wall fractures. ...
- Deep tissue injuries. ...
- Atrial fibrillation. ...
- Phlebitis and thrombophlebitis. ...
- Embolism and thrombosis. ...
- Poisoning, adverse effects and underdosing. ...
- Heatstroke. ...
- Legal intervention.
How do I find diagnosis codes?
WHO Updates ICD-10 codes?
WHO still uses ICD-9 codes?
What is the difference between ICD-9 and ICD-9-CM?
When did ICD-10 replace ICD-9?
Known As
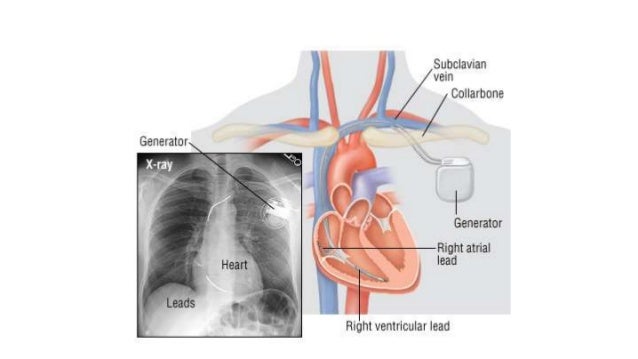
Defibrillator is also known as automatic implantable cardiac defibrillator, biventricular automatic implantable cardioverter defibrillator present, biventricular implanted defibrillator normal function, cardiac defibrillator in situ, presence of AICD, presence of biventricular AICD, presence of biventricular implantable cardiac defibrillator, and presence of cardiac defibrillator..
Defibrillator Definition and Symptoms
A defibrillator is a medical device that is used to “jump start” a person’s heart using electrical shock. The different types of defibrillators are automated external defibrillator (AED) and sudden cardiac arrest (SCA). Defibrillators are used for cardiac arrest, heart attacks, and if the heart stops working properly.
Does Medicare cover ICDs for cardiac death?
Although Medicare’s NCD provides coverage criteria for primary and secondary prevention of sudden death, the coverage of the two indications is not the same.#N#Secondary prevention of sudden death is covered, as long as there are no contraindications. Medicare only covers ICDs placed for the primary prevention of sudden cardiac death if data is submitted to an FDA-approved category B IDE clinical trial, a trial under the CMS Clinical Trial Policy, or a qualifying data collection system, including approved clinical trials and registries. CMS selected the American College of Cardiology’s National Cardiovascular Data Registry (ACC-NCDR) ICD Registry as the mandated national registry for ICDs in October 2005. The ClinicalTrials.gov identifier for this data repository is NCT01999140.#N#When an ICD is implanted in the outpatient setting, providers notify Medicare that data is being submitted to the ACC-NCDR, as required in the NCD, by appending modifier Q0 Investigational clinical service provided in a clinical research study that is in an approved clinical research study to the CPT® code for the implantation procedure. If the performed procedure is for primary prevention of sudden death, and modifier Q0 is not appended, there is no coverage for the ICD implantation.#N#Modifier Q0 is required only when the ICD is implanted for primary prevention of sudden death. Modifier Q0 may be appended to claims for secondary prevention indications (those meeting non-restricted coverage requirements due to clinical indications) when data is being submitted to a data collection system.#N#Although the ACC-NCDR may not meet the expected definition of a clinical trial, the use of modifier Q0 categorizes the procedure as part of a clinical study. When a clinical study is performed, ICD-10-CM Z00.6 Encounter for examination for normal comparison and control in clinical research program typically is required in either the primary or secondary position on the claim.#N#The following items are required when billing for ICDs implanted for primary prevention of sudden death:#N#For hospital outpatient billing, clinical trial registry number 1999140 in the electronic claim equivalent, 837I (Loop 2300 REF02 (REF01=P4) for an 837I claim when a clinical trial claim includes:
Does Medicare cover ICDs?
Medicare only covers ICDs placed for the primary prevention of sudden cardiac death if data is submitted to an FDA-approved category B IDE clinical trial, a trial under the CMS Clinical Trial Policy, or a qualifying data collection system, including approved clinical trials and registries.
What is the primary prevention of sudden cardiac death?
The coverage falls into two major categories: primary prevention of sudden cardiac death and secondary prevention of sudden cardiac death. Primary prevention ICD implantation is to prevent sudden death in a patient who has poor cardiac function and potentially abnormal heart rhythm that could lead to cardiac arrest.
Who is Ruth Broek?
Ruth Broek, MBA, CIRCC, COC, CCS, RTR, CHC, vice president of ZHealth Publishing, is responsible for all support functions, including researching coverage issues, maintaining coding policies, keeping the reference library updated, and maintaining website information. She also performs chargemaster reviews and revenue cycle process improvement for facilities across the country. Broek is a member of the Murfreesboro, Tenn., local chapter.
Popular Posts:
- 1. icd 10 code for gustillo iii tib fib fracture
- 2. icd 10 cm code for t acute respiratory failure due to severe viral sepsis
- 3. icd 10 code for transportation problems
- 4. icd-10 code for nexplanon status
- 5. icd 10 code for personal history sickle cell disease
- 6. icd 9 code for wound to left alrm
- 7. icd 10 code for right periorbital hematoma
- 8. icd 10 code for tendenitios
- 9. icd 10 diagnosis code for muscle spasm
- 10. icd 9 code for dtap vaccine