| CPT® (CDT codes and descriptions are copyright American Dental Association) | |
|---|---|
| 86900 | |
| Z94.0 | Kidney transplant status |
| Z94.4 | Liver transplant status |
| ICD-9-CM | |
What is JAK2 gene mutation?
This amino acid change results in a JAK2 protein that is constantly "on," leading to uncontrolled blood cell production. Other mutations in the JAK2 gene are also associated with MPNs.
What does a positive Jak2 V617F mutation test mean?
A positive JAK2 V617F mutation test, along with other supporting clinical signs, means it is likely that the person tested has an MPN. Other testing, such as a bone marrow biopsy, may need to be performed to determine which MPN the person has and to evaluate its severity.
What are the tests for JAK2 mutations that lead to MPNS?
The primary genetic test for JAK2 mutations that lead to MPNs is JAK2 V617F, named for a mutation at a specific location in the JAK2 gene. It is typically ordered first. If it is negative, then tests for other mutations in the JAK2 gene that are also associated with MPNs, such as JAK2 exon 12, may be used to help make a diagnosis.
What is the sensitivity of the JAK2 test?
Other mutations that may occur in the JAK2 gene will not be detected. In vitro studies have indicated that this assay has an analytical sensitivity of 1%. This test was developed, and its performance characteristics determined, by LabCorp. It has not been cleared or approved by the US Food and Drug Administration (FDA).
What is JAK2 kinase mutation?
In JAK2, this kind of mutation, called a point mutation, replaces the normal amino acid valine (abbreviated V) with phenylalanine (abbreviated F). This amino acid change results in a JAK2 protein that is constantly “on,” leading to uncontrolled blood cell production.
Is JAK2 a leukemia mutation?
JAK2 mutations are rare in de novo acute myeloid leukemia (AML), and JAK2-mutated acute myeloid leukemia (AML) patients usually have a previous history of myeloproliferative neoplasms (MPNs).
What is JAK2 positive myeloproliferative disorder?
Mutations in this gene result in constant increased blood cell production. Depending on which cells are stimulated by a JAK2 mutation, one of several types of myeloproliferative disorders can develop, including polycythemia vera, essential thrombocythemia, and primary myelofibrosis.
Does Medicare cover JAK2?
For laboratories performing next generation sequencing (NGS or "hotspot") testing platforms: Molecular testing for BCR-ABL, JAK 2, JAK, exon 12, and CALR/MPL genes by NGS is covered as medically necessary for the identification of myeloproliferative disorders.
What is the CPT code for JAK2 V617F mutation analysis?
CPT Codes*: JAK2 V617F Only: 81270 JAK2 V617F and EXON 12-14 run concurrently or by reflex: 81270, 81403.
What is wild type JAK2?
The JAK2 Wild Type Reference Standard is a highly-characterized, biologically-relevant quality control material used to assess the performance of assays that detect somatic mutations, such as Sanger and qPCR sequencing assays.
What is the ICD 10 code for myeloproliferative disorder?
1.
What is JAK2 thrombocythemia?
[4] According to the World Health Organization, essential thrombocytosis is a disease that occurs when the platelet count is more than 450000 with the presence of Janus kinase 2 (JAK2), Calreticulin (CALR) or myeloproliferative leukemia virus oncogene (MPL) mutation, lacking clonal or reactive causes.
How do you get JAK2 mutation?
Rather than being inherited, JAK2 V617F mutations are acquired, occurring at random. As they occur in cells that do not produce eggs or sperm, the mutation is not passed on to a person's children.
Is CPT code 81206 genetic testing?
CPT® 81206, Under Genetic Analysis Procedures The Current Procedural Terminology (CPT®) code 81206 as maintained by American Medical Association, is a medical procedural code under the range - Genetic Analysis Procedures.
Is CPT 81291 covered by Medicare?
HMO, PPO, Individual Marketplace, & Elite/ProMedica Medicare Plan MTHFR (methyltetrahydrofolate reductase) gene testing (81291) is non-covered. Advantage Genetic testing for hereditary thrombophilia (81291) requires prior authorization.
Is CPT code 81479 genetic testing?
Unlisted Molecular Pathology - CPT Code 81479 However, when reporting CPT code 81479, the specific gene being tested must be entered in block 80 (Part A for the UBO4 claim), box 19 (Part B for a paper claim) or electronic equivalent of the claim.
Popular Posts:
- 1. icd 10 code for pulmonary nodule
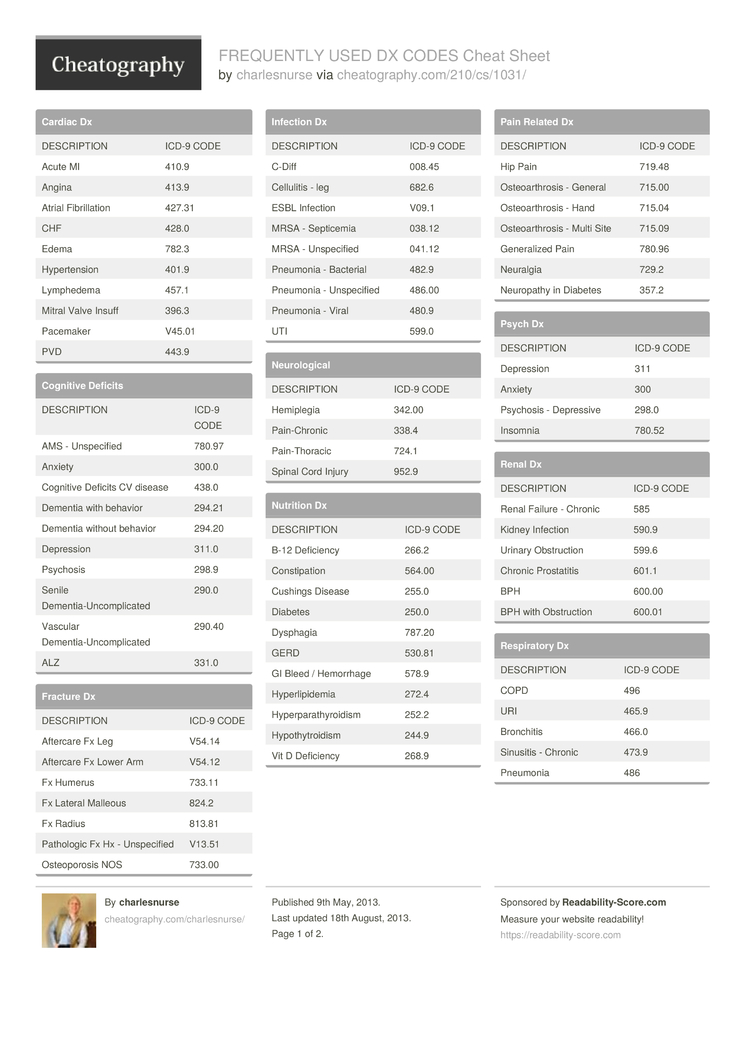
- 2. what is the icd-10 code for generalized pain
- 3. icd 10 code for iron level
- 4. icd 10 code for thrombocytosis
- 5. icd 10 code for nondisplaced fracture of the fifth middle phalanx
- 6. icd 10 cm code for encounter for chemotherapy for malignant glioma of the frontal lobe
- 7. icd 10 code for diabetic foot ulcer infection
- 8. icd 10 code for insulin pump
- 9. icd 10 code for colostomy hemorrhage
- 10. icd 10 code for high creatinine levels