What is the billing code for Botox?
BOTOX 200 U nit vial 00023 -3921-02 . Providers should submit the appropriate charges for the number of Botox units used (not number of vials) using the specific HCPCS II code J0585- Injection, onabotulinumtoxinA, 1 unit).
What is the administration code for Botox?
If so, the administration of the botox is included in the procedure code. I don't believe you should use either. 90471 is strictly for immunizations. 90772 is used for therapeutic, prophylactic, or diagnostic, which are not the cases either. I think you would use 11950 - 11954. These are just introduction codes.
What is the CPT code for administration of Botox injection?
General Guidelines
- Use the appropriate Healthcare Common Procedure Coding System (HCPCS) based on code descriptor.
- Not Otherwise Classified (NOC) codes should only be reported for those drugs that do not have a valid HCPCS code which describes the drug being administered.
- Remarks are required to include dosage, name of drug, and route of administration.
What is the J code for Botox?
HCPCS J-Codes Drugs administered other than oral method, chemotherapy drugs
- J0120 Injection, tetracycline, up to 250 mg
- J0121 Injection, omadacycline, 1 mg
- J0122 Injection, eravacycline, 1 mg
- J0129 Injection, abatacept, 10 mg (code may be used for medicare when drug administered under the direct supervision of a physician, not for use when drug is self administered)
- J0130 Injection abciximab, 10 mg
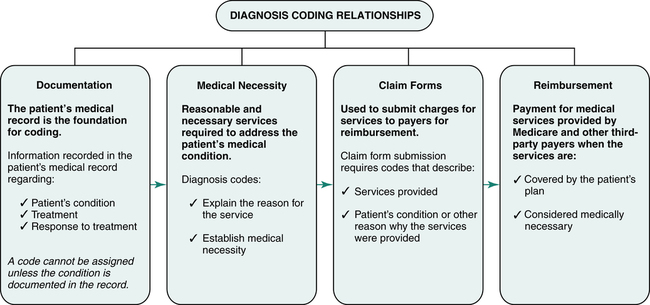
What is the ICD 10 code for cosmetic Botox?
The ICD-10-CM code that should be filed in this situation is Z41. 1, "Encounter for cosmetic surgery."
What is the ICD 10 code for Encounter for cosmetic surgery?
ICD-10 Code for Encounter for cosmetic surgery- Z41. 1- Codify by AAPC.
What is diagnosis code Z71 89?
Other specified counselingICD-10 code Z71. 89 for Other specified counseling is a medical classification as listed by WHO under the range - Factors influencing health status and contact with health services .
What is the ICD 10 code for facial?
Disorder of facial nerve, unspecified G51. 9 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes. The 2022 edition of ICD-10-CM G51. 9 became effective on October 1, 2021.
What is the ICD 10 code for cosmetic?
Z41. 1 - Encounter for cosmetic surgery. ICD-10-CM.
What does cosmetic surgery mean?
Cosmetic Surgery: Focused on Enhancing Appearance Improving aesthetic appeal, symmetry, and proportion are the key goals. An aesthetic surgery can be performed on all areas of the head, neck, and body. Since cosmetic procedures treat areas that function properly, cosmetic surgery is designated as elective.
Can Z76 89 be a primary diagnosis?
The patient's primary diagnostic code is the most important. Assuming the patient's primary diagnostic code is Z76. 89, look in the list below to see which MDC's "Assignment of Diagnosis Codes" is first. That is the MDC that the patient will be grouped into.
What is the ICD-10 code for injection?
ICD-10 code T80 for Complications following infusion, transfusion and therapeutic injection is a medical classification as listed by WHO under the range - Injury, poisoning and certain other consequences of external causes .
What is I10 diagnosis?
ICD-Code I10 is a billable ICD-10 code used for healthcare diagnosis reimbursement of Essential (Primary) Hypertension.
What is the ICD 10 code for facial aging?
The 2022 edition of ICD-10-CM L98. 8 became effective on October 1, 2021. This is the American ICD-10-CM version of L98.
What is the ICD 10 code for skin lesion?
ICD-10-CM Code for Disorder of the skin and subcutaneous tissue, unspecified L98. 9.
What is the ICD 10 code for right facial droop?
ICD-10-CM Code for Facial weakness R29. 810.
What are the most common adverse reactions to Botox?
The most frequently reported adverse reactions (3-10% of adult patients) following injection of BOTOX in double-blind studies included injection site pain and hemorrhage, non-axillary sweating, infection, pharyngitis, flu syndrome, headache, fever, neck or back pain, pruritus, and anxiety.
What is the potency of botox?
The potency Units of BOTOX (onabotulinumtoxinA) for injection are specific to the preparation and assay method utilized. They are not interchangeable with other preparations of botulinum toxin products and, therefore, units of biological activity of BOTOX cannot be compared to nor converted into units of any other botulinum toxin products assessed with any other specific assay method [see Warnings and Precautions
What is Botox used for?
BOTOX® is indicated for the treatment of lower limb spasticity in adult patients to decrease the severity of increased muscle tone in ankle and toe flexors (gastrocnemius, soleus, tibialis posterior, flexor hallucis longus, and flexor digitorum longus).
What is the discontinuation rate for Botox?
In double-blind, placebo-controlled chronic migraine efficacy trials (Study 1 and Study 2), the discontinuation rate was 12% in the BOTOX treated group and 10% in the placebo-treated group. Discontinuations due to an adverse event were 4% in the BOTOX group and 1% in the placebo group. The most frequent adverse events leading to discontinuation in the BOTOX group were neck pain, headache, worsening migraine, muscular weakness and eyelid ptosis.
How many units of Botox are in the eye?
In a study of blepharospasm patients who received an average dose per eye of 33 Units (injected at 3 to 5 sites) of the currently manufactured BOTOX, the most frequently reported adverse reactions were ptosis (21%), superficial punctate keratitis (6%), and eye dryness (6%).
How long should I wait to shave my arm before a hyperhidrosis test?
Patients should shave underarms and abstain from use of over-the-counter deodorants or antiperspirants for 24 hours prior to the test. Patient should be resting comfortably without exercise, hot drinks for approximately 30 minutes prior to the test. Dry the underarm area and then immediately paint it with iodine solution. Allow the area to dry, then lightly sprinkle the area with starch powder. Gently blow off any excess starch powder. The hyperhidrotic area will develop a deep blue-black color over approximately 10 minutes.
Why are adverse reactions not reflected in clinical practice?
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
General Information
CPT codes, descriptions and other data only are copyright 2020 American Medical Association. All Rights Reserved. Applicable FARS/HHSARS apply.
Article Guidance
This article contains coding or other guidelines that complement the local coverage determination (LCD) for Botulinum Toxins. Coding Information: Procedure codes may be subject to National Correct Coding Initiative (NCCI) edits or OPPS packaging edits.
Bill Type Codes
Contractors may specify Bill Types to help providers identify those Bill Types typically used to report this service. Absence of a Bill Type does not guarantee that the article does not apply to that Bill Type.
Revenue Codes
Contractors may specify Revenue Codes to help providers identify those Revenue Codes typically used to report this service. In most instances Revenue Codes are purely advisory. Unless specified in the article, services reported under other Revenue Codes are equally subject to this coverage determination.
How long does a migraine last with a botox injection?
BOTOX® (onabotulinumtoxinA) for injection is indicated for the prophylaxis of headaches in adult patients with chronic migraine (≥ 15 days per month with headache lasting 4 hours a day or longer).
Can botulinum toxin cause death?
There have been spontaneous reports of death, sometimes associated with dysphagia, pneumonia, and/or other significant debility or anaphylaxis, after treatment with botulinum toxin. There have also been reports of adverse events involving the cardiovascular system, including arrhythmia and myocardial infarction, some with fatal outcomes. Some of these patients had risk factors including cardiovascular disease. The exact relationship of these events to the botulinum toxin injection has not been established.
Is Botox contraindicated?
BOTOX® is contraindicated in the presence of infection at the proposed injection site(s) and in individuals with known hypersensitivity to any botulinum toxin preparation or to any of the components in the formulation.
:max_bytes(150000):strip_icc()/GettyImages-946399418-5c01f4fc46e0fb0001dc690f.jpg)
Popular Posts:
- 1. icd-10 code for boil
- 2. icd 10 code for neoplastic pain
- 3. icd 10 cm code for post c3-c4 fusion
- 4. icd 10 code for dense mass tissue
- 5. icd 10 code for schizoaffective disorder depressive type
- 6. icd 10 code for nasolacrimal duct obstruction
- 7. icd 9 code for diverticular disease
- 8. icd 10 code for chondrocalcinosis
- 9. icd 10 code for hallux cellulitis
- 10. icd-10 code for initial prescrption for brth control