What to expect when getting tested for hepatitis C?
The CDC recommend one-time screening for:
- anyone who has used injected drugs, especially if they shared any equipment
- people with certain health issues, such as those: with alanine aminotransferase levels outside the normal range who have had maintenance hemodialysis who have had organ transplants or blood transfusions
- children whose birth parent has hepatitis C
What is the diagnosis code for hepatitis screening?
- You’re at high risk because you use or have used illicit injection drugs.
- You had a blood transfusion before 1992.
- You were born between 1945-1965.
What is the ICD 10 diagnosis code for?
The ICD-10-CM is a catalog of diagnosis codes used by medical professionals for medical coding and reporting in health care settings. The Centers for Medicare and Medicaid Services (CMS) maintain the catalog in the U.S. releasing yearly updates.
Do you need to get screened for hepatitis C?
Most people who have hepatitis C do not show symptoms, so the Centers for Disease Control and Prevention (CDC) recommends that anyone who falls in the high-risk age range get tested. Hepatitis C is caused by a virus that attacks the liver. Its effects can be serious and long-lasting.
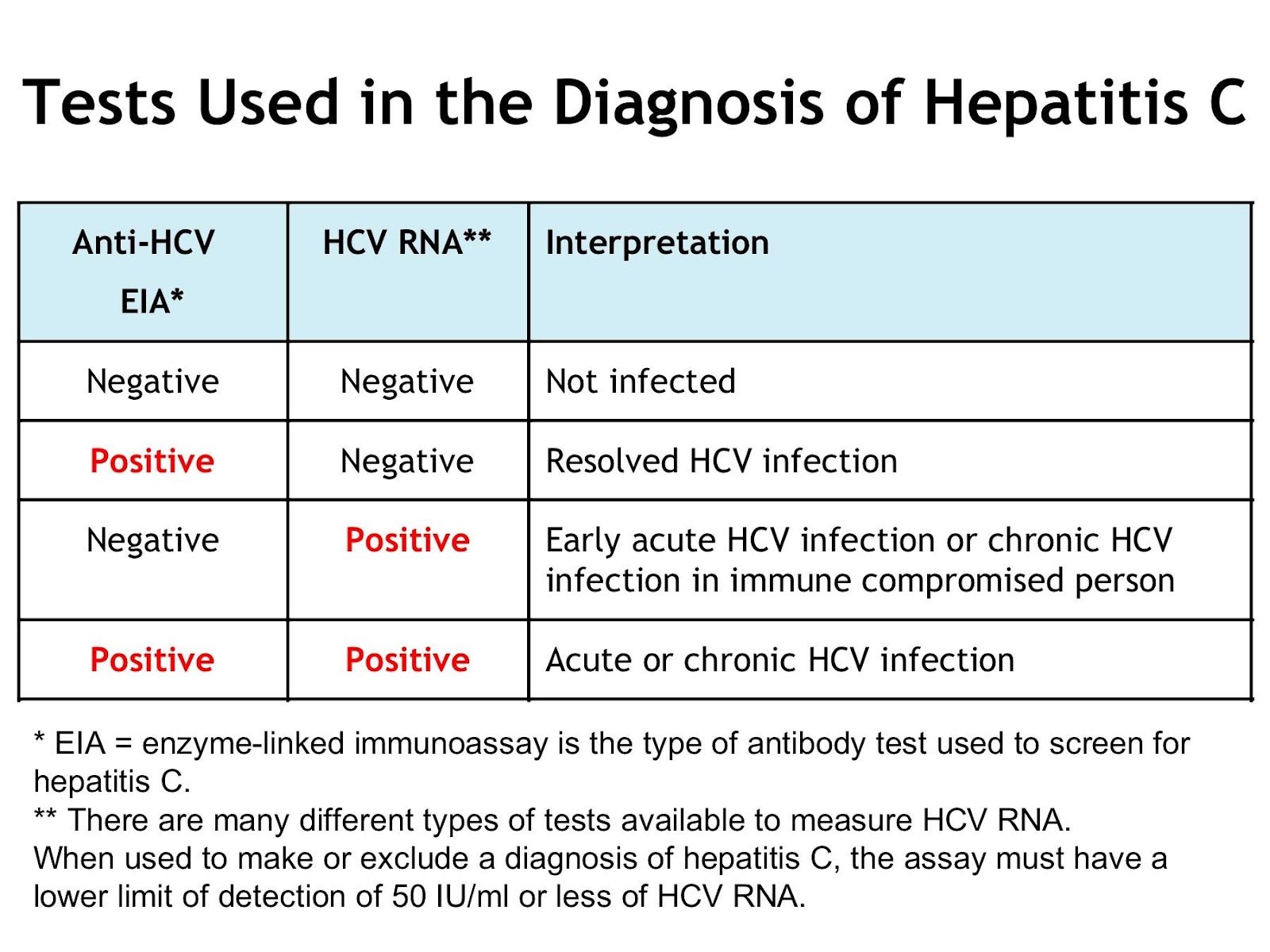
What is the ICD-10 code for hepatitis screening?
2022 ICD-10-CM Diagnosis Code Z11. 59: Encounter for screening for other viral diseases.
What ICD-10 code covers hepatitis panel?
ICD-10-CM Diagnosis Code B18 B18.
What is diagnosis code z1159?
For asymptomatic individuals who are being screened for COVID-19 and have no known exposure to the virus, and the test results are either unknown or negative, assign code Z11. 59, Encounter for screening for other viral diseases.
What is the screening test for hepatitis B and C?
What is a hepatitis panel? Hepatitis is a type of liver disease. Viruses called hepatitis A, hepatitis B, and hepatitis C are the most common causes of hepatitis. A hepatitis panel is a blood test that checks to see if you have a hepatitis infection caused by one of these viruses.
How do you code hepatitis?
Assign code 070.1 for a hepatitis A diagnosis or 070.0 for hepatitis A with hepatic coma. 070.32, Chronic hepatitis B without hepatic coma. 070.71, Unspecified viral hepatitis C with hepatic coma.
What covers a hepatitis panel?
This panel consists of the following tests: Hepatitis A antibody (HAAb), IgM Antibody; Hepatitis B core antibody (HBcAb), IgM Antibody; Hepatitis B surface antigen (HBsAg); and.
What is the ICD-10 code for hepatitis?
Unspecified viral hepatitis without hepatic coma B19. 9 is a billable/specific ICD-10-CM code that can be used to indicate a diagnosis for reimbursement purposes. The 2022 edition of ICD-10-CM B19. 9 became effective on October 1, 2021.
When do you use DX Z11 52?
These codes are therefore an assumption of how it was likely being reported. Note: The new code Z11. 52 (Encounter for screening for COVID-19) effective 1/1/21, is not appropriate for screening until the pandemic is declared over (per January 2021 updated guidelines I.C.
When do you use ICD Z11 52?
For example, Z11. 52 will replace Z11. 59 (Encounter for screening for other viral diseases), which the CDC previously said should be used when patients being screened for COVID-19 have no symptoms, no known exposure to the virus, and test results that are either unknown or negative.
What is the labcorp test code for hepatitis panel?
144025: Viral Hepatitis Screening and Diagnosis (HBV, HCV) | Labcorp.
What should I order for hepatitis B screening?
There is a simple hepatitis B blood test that your doctor or health clinic can order called the “Hepatitis B Panel”. Only one blood sample is needed, but there are 3 parts to the hepatitis B panel.
What is hep be Ab?
anti-HBs or HBsAb (Hepatitis B surface antibody) - A "positive" or "reactive" anti-HBs (or HBsAb) test result indicates that a person is protected against the hepatitis B virus. This protection can be the result of receiving the hepatitis B vaccine or successfully recovering from a past hepatitis B infection.
Hepatitis A,B,C,D,E and Liver Diseases
Hepatitis refer to inflammation of the liver that can caused by virus infections that affect the liver. Viral hepatitis is a relatively common disease (25 patients in every 100,000 citizens in the US) caused by a diverse group of hepatotropic agents that lead to liver inflammation and liver cell death.
CPT Codes for Hepatitis C, B Screening Test
The CDC has recommendations regarding Hepatitis screening for both hepatitis B and hepatitis C for persons at high risk for infection.
Conditions for Coverage of HBsAg Serologic Testing
The patient’s primary care physician or practitioner, an eligible Medicare provider, must order the screening within the context of a primary care setting.
Diagnosis Coding for HBV Screening
For HBV screening in pregnant women (CPT codes 86704, 86706, 87340 and 87341) report Z11.59 Encounter for screening for other viral diseases with one of the following diagnosis codes:
Popular Posts:
- 1. icd 10 code for asherman's syndrome
- 2. icd 10 code for right hip injury
- 3. icd-10-cm code for overactive bladder
- 4. icd 10 code for fracture right foot
- 5. icd 10 code for left eye prosthesis
- 6. icd 9 code for diabetes 251.2
- 7. icd 10 code for anoxic leukocytosis
- 8. icd 10 code for chronic renal disease stage 3
- 9. what is the correct icd 10 code for n938
- 10. icd 10 code for paroxysmal supraventicular tachycardia